PPARδ signaling mediates the cytotoxicity of DHA in H9c2 cells
- PMID: 25300478
- PMCID: PMC4486642
- DOI: 10.1016/j.toxlet.2014.09.029
PPARδ signaling mediates the cytotoxicity of DHA in H9c2 cells
Abstract
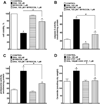
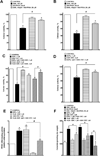
Docosahexaenoic acid (22:6n3, DHA) is an n-3 polyunsaturated fatty acid (PUFA) known to affect numerous biological functions. While DHA possesses many properties that impact cell survival such as suppressing cell growth and inducing apoptosis, the exact molecular and cellular mechanism(s) remain unknown. Peroxisome proliferator-activated receptors (PPARs) are a family of nuclear receptors that regulate many cell pathways including cell death. As DHA acts as a ligand to PPARs the aim of this study was to examine the involvement of PPARδ in DHA-mediated cytotoxicity toward H9c2 cells. Treatment with DHA (100μM) resulted in a significant decline in cell viability, cellular metabolic activity and total antioxidant capacity coinciding with increased total proteasome activities and activity of released lactate dehydrogenase (LDH). No changes in reactive oxygen species (ROS) production or accumulation of lipid peroxidation products were observed but DHA promoted apoptotic cell death as detected by flow cytometry, increased caspase-3 activity and decreased phosphorylation of Akt. Importantly, DHA enhanced PPARδ DNA binding activity in H9c2 cells strongly signifying that the cytotoxic effect of DHA might be mediated via PPARδ signaling. Co-treatment with the selective PPARδ antagonist GSK 3787 (1μM) abolished the cytotoxic effects of DHA in H9c2 cells. Cytotoxic effects of DHA were attenuated by co-treatment with myriocin, a selective inhibitor of serine palmitoyl transferase (SPT), preventing de novo ceramide biosynthesis. LC/MS analysis revealed that treatment with DHA resulted in the accumulation of ceramide, which was blocked by GSK 3787. Interestingly, inhibition of cytochrome P450 (CYP) oxidase with MS-PPOH (50μM) abolished DHA-mediated cytotoxicity suggesting downstream metabolites as the active mediators. We further demonstrate that CYP oxidase metabolites of DHA, methyl epoxy docosapentaenoate (EDP methyl esters, 1μM) (mix 1:1:1:1:1:1; 4,5-, 7,8-, 10,11-, 13,14-, 16,17- and 19,20-EDP methyl esters) and 19,20-EDP cause cytotoxicity via activation of PPARδ signaling leading to increased levels of intracellular ceramide. These results illustrate novel pathways for DHA-induced cytotoxicity that suggest an important role for CYP-derived metabolites, EDPs.
Keywords: Apoptosis; Ceramide; Docosahexaenoic acid; Epoxydocosapentaenoic acids; H9c2 cells; PPARδ.
Copyright © 2014 Elsevier Ireland Ltd. All rights reserved.
Conflict of interest statement
No conflict of interests declared.
Figures
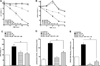
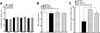

Similar articles
-
DHA and 19,20-EDP induce lysosomal-proteolytic-dependent cytotoxicity through de novo ceramide production in H9c2 cells with a glycolytic profile.Cell Death Discov. 2018 Aug 20;4:29. doi: 10.1038/s41420-018-0090-1. eCollection 2018. Cell Death Discov. 2018. PMID: 30131878 Free PMC article.
-
Differential responses to docosahexaenoic acid in primary and immortalized cardiac cells.Toxicol Lett. 2013 Jun 7;219(3):288-97. doi: 10.1016/j.toxlet.2013.03.010. Epub 2013 Mar 21. Toxicol Lett. 2013. PMID: 23523905
-
Peroxisome proliferator-activated receptor delta agonists attenuated the C-reactive protein-induced pro-inflammation in cardiomyocytes and H9c2 cardiomyoblasts.Eur J Pharmacol. 2010 Sep 15;643(1):84-92. doi: 10.1016/j.ejphar.2010.06.031. Epub 2010 Jun 28. Eur J Pharmacol. 2010. PMID: 20599914
-
CYP-eicosanoids--a new link between omega-3 fatty acids and cardiac disease?Prostaglandins Other Lipid Mediat. 2011 Nov;96(1-4):99-108. doi: 10.1016/j.prostaglandins.2011.09.001. Epub 2011 Sep 16. Prostaglandins Other Lipid Mediat. 2011. PMID: 21945326 Review.
-
Cytochrome P450-derived eicosanoids and heart function.Pharmacol Ther. 2017 Nov;179:47-83. doi: 10.1016/j.pharmthera.2017.05.005. Epub 2017 May 25. Pharmacol Ther. 2017. PMID: 28551025 Review.
Cited by
-
SIRT Is Required for EDP-Mediated Protective Responses toward Hypoxia-Reoxygenation Injury in Cardiac Cells.Front Pharmacol. 2016 May 17;7:124. doi: 10.3389/fphar.2016.00124. eCollection 2016. Front Pharmacol. 2016. PMID: 27242531 Free PMC article.
-
Cancer Prevention and Therapy with Polyphenols: Sphingolipid-Mediated Mechanisms.Nutrients. 2018 Jul 21;10(7):940. doi: 10.3390/nu10070940. Nutrients. 2018. PMID: 30037082 Free PMC article. Review.
-
Ethanol and C2 ceramide activate fatty acid oxidation in human hepatoma cells.Sci Rep. 2018 Aug 27;8(1):12923. doi: 10.1038/s41598-018-31025-0. Sci Rep. 2018. PMID: 30150688 Free PMC article.
-
PPARδ, a Potential Therapeutic Target for Heart Disease.Nucl Receptor Res. 2018;5:101375. doi: 10.32527/2018/101375. Epub 2018 Oct 30. Nucl Receptor Res. 2018. PMID: 31032335 Free PMC article.
-
Cadmium Toxicity Is Regulated by Peroxisome Proliferator-Activated Receptor δ in Human Proximal Tubular Cells.Int J Mol Sci. 2022 Aug 3;23(15):8652. doi: 10.3390/ijms23158652. Int J Mol Sci. 2022. PMID: 35955783 Free PMC article.
References
-
- Abe K, Matsuki N. Measurement of cellular 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction activity and lactate dehydrogenase release using MTT. Neurosci. Res. 2000;38:325–329. - PubMed
-
- Al-Gubory KH. Mitochondria: omega-3 in the route of mitochondrial reactive oxygen species. Int. J. Biochem. Cell Biol. 2012;44:1569–1573. - PubMed
-
- Barak Y, Nelson MC, Ong ES, Jones YZ, Ruiz-Lozano P, Chien KR, Koder A, Evans RM. PPAR gamma is required for placental, cardiac, and adipose tissue development. Mol. Cell. 1999;4:585–595. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

