Belonging to a network--microRNAs, extracellular vesicles, and the glioblastoma microenvironment
- PMID: 25301812
- PMCID: PMC4482852
- DOI: 10.1093/neuonc/nou292
Belonging to a network--microRNAs, extracellular vesicles, and the glioblastoma microenvironment
Abstract
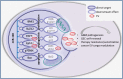
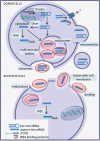
The complexity of glioblastoma multiforme (GBM) and its distinct pathophysiology belong to a unique brain microenvironment and its cellular interactions. Despite extensive evidence of a role for microRNAs in GBM cells, little is known about microRNA-dependent communication between different cellular compartments of the microenvironment that may contribute to the tumor phenotype. While the majority of microRNAs are found intracellularly, a significant number of microRNAs have been observed outside of cells, often encapsulated in secreted extracellular vesicles (EVs). The function of these circulating/secreted microRNAs has not been explored in the context of the brain tumor microenvironment. Establishing how microRNAs are involved in the regulation of oncogenic signaling networks between tumor cells and stroma is likely to add a needed additional layer of complexity to the tumor network, consisting of intercellular communication. More importantly, microRNA/EV signaling may provide an additional therapeutic target for this deadly disease.
Keywords: exosomes; extracellular vesicles; glioblastoma multiforme; microRNA; tumor microenvironment.
© The Author(s) 2014. Published by Oxford University Press on behalf of the Society for Neuro-Oncology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures


References
-
- Hoelzinger DB, Demuth T, Berens ME. Autocrine factors that sustain glioma invasion and paracrine biology in the brain microenvironment. J Natl Cancer Inst. 2007;99(21):1583–1593. - PubMed
-
- Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer. 2004;4(1):71–78. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

