Impact of combination antiretroviral therapy in the NOD.c3c4 mouse model of autoimmune biliary disease
- PMID: 25302564
- PMCID: PMC4403978
- DOI: 10.1111/liv.12699
Impact of combination antiretroviral therapy in the NOD.c3c4 mouse model of autoimmune biliary disease
Abstract
Background & aims: The NOD.c3c4 mouse model develops autoimmune biliary disease characterized by spontaneous granulomatous cholangitis, antimitochondrial antibodies and liver failure. This model for primary biliary cirrhosis (PBC) has evidence of biliary infection with mouse mammary tumour virus (MMTV), suggesting that the virus may have a role in cholangitis development and progression of liver disease in this mouse model. We tested the hypothesis that MMTV infection is associated with cholangitis in the NOD.c3c4 mouse model by investigating whether antiretroviral therapy impacts on viral levels and liver disease.
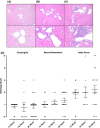
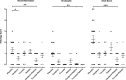
Methods: NOD.c3c4 mice were treated with combination antiretroviral therapy. Response to treatment was studied by measuring MMTV RNA in the liver, liver enzyme levels in serum and liver histology using a modified Ishak score.
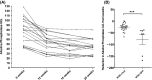
Results: Combination therapy with the reverse transcriptase inhibitors, tenofovir and emtricitabine, resulted in a significant reduction in serum liver enzyme levels, attenuation of cholangitis and decreased MMTV levels in the livers of NOD.c3c4 mice. Furthermore, treatment with the retroviral protease inhibitors, lopinavir and ritonavir, in addition to the reverse transcriptase inhibitors, resulted in further decrease in MMTV levels and attenuation of liver disease in this model.
Conclusions: The attenuation of cholangitis with regimens containing the reverse transcriptase inhibitors, tenofovir and emtricitabine, and the protease inhibitors, lopinavir and ritonavir, suggests that retroviral infection may play a role in the development of cholangitis in this model.
Keywords: NOD.c3c4; autoimmune biliary disease; combination antiretroviral therapy; mouse mammary tumour virus; primary biliary cirrhosis.
© 2014 The Authors. Liver International Published by John Wiley & Sons Ltd.
Figures



Similar articles
-
Combination antiretroviral studies for patients with primary biliary cirrhosis.World J Gastroenterol. 2016 Jan 7;22(1):349-60. doi: 10.3748/wjg.v22.i1.349. World J Gastroenterol. 2016. PMID: 26755881 Free PMC article. Review.
-
Mouse mammary tumor virus in anti-mitochondrial antibody producing mouse models.J Hepatol. 2011 Oct;55(4):876-84. doi: 10.1016/j.jhep.2011.01.037. Epub 2011 Feb 18. J Hepatol. 2011. PMID: 21334408
-
NOD.c3c4 congenic mice develop autoimmune biliary disease that serologically and pathogenetically models human primary biliary cirrhosis.J Exp Med. 2006 May 15;203(5):1209-19. doi: 10.1084/jem.20051911. Epub 2006 Apr 24. J Exp Med. 2006. PMID: 16636131 Free PMC article.
-
Primary biliary cirrhosis: new thoughts on pathophysiology and treatment.Curr Gastroenterol Rep. 2002 Feb;4(1):45-51. doi: 10.1007/s11894-002-0037-8. Curr Gastroenterol Rep. 2002. PMID: 11825541 Review.
-
Linking human beta retrovirus infection with primary biliary cirrhosis.Gastroenterol Clin Biol. 2010 Aug-Sep;34(6-7):359-66. doi: 10.1016/j.gcb.2010.04.014. Epub 2010 Jun 30. Gastroenterol Clin Biol. 2010. PMID: 20580176 Review.
Cited by
-
Apples to Apples? A Comparison of Real-World Tolerability of Antiretrovirals in Patients with Human Immunodeficiency Virus Infection and Patients with Primary Biliary Cholangitis.Viruses. 2022 Mar 3;14(3):516. doi: 10.3390/v14030516. Viruses. 2022. PMID: 35336923 Free PMC article. Review.
-
Isolation of a Human Betaretrovirus from Patients with Primary Biliary Cholangitis.Viruses. 2022 Apr 24;14(5):886. doi: 10.3390/v14050886. Viruses. 2022. PMID: 35632628 Free PMC article.
-
Novel therapeutic targets in primary biliary cirrhosis.Nat Rev Gastroenterol Hepatol. 2015 Mar;12(3):147-58. doi: 10.1038/nrgastro.2015.12. Epub 2015 Feb 3. Nat Rev Gastroenterol Hepatol. 2015. PMID: 25645973 Review.
-
Endogenous antigens shape the transcriptome and TCR repertoire in an autoimmune arthritis model.J Clin Invest. 2024 Nov 26;135(2):e174647. doi: 10.1172/JCI174647. J Clin Invest. 2024. PMID: 39589811 Free PMC article.
-
Combination antiretroviral studies for patients with primary biliary cirrhosis.World J Gastroenterol. 2016 Jan 7;22(1):349-60. doi: 10.3748/wjg.v22.i1.349. World J Gastroenterol. 2016. PMID: 26755881 Free PMC article. Review.
References
-
- Koarada S, Wu Y, Fertig N, et al. Genetic control of autoimmunity: protection from diabetes, but spontaneous autoimmune biliary disease in a nonobese diabetic congenic strain. J Immunol. 2004;173:2315–23. - PubMed
-
- Oertelt S, Lian ZX, Cheng CM, et al. Anti-mitochondrial antibodies and primary biliary cirrhosis in TGF-beta receptor II dominant-negative mice. J Immunol. 2006;177:1655–60. - PubMed
-
- Salas JT, Banales JM, Sarvide S, et al. Ae2a, b-deficient mice develop antimitochondrial antibodies and other features resembling primary biliary cirrhosis. Gastroenterology. 2008;134:1482–93. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

