CD74 interferes with the expression of fas receptor on the surface of lymphoma cells
- PMID: 25304249
- PMCID: PMC4210479
- DOI: 10.1186/s13046-014-0080-y
CD74 interferes with the expression of fas receptor on the surface of lymphoma cells
Abstract
Background: Resistance to Fas-mediated apoptosis limits the efficacy of currently available chemotherapy regimens. We identified CD74, which is known to be overexpressed in hematological malignancies, as one of the factors interfering with Fas-mediated apoptosis.
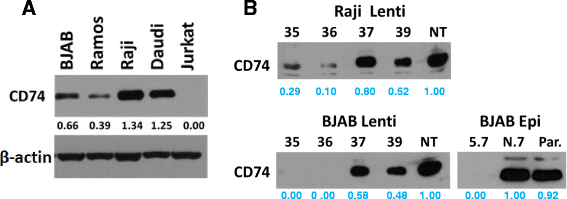
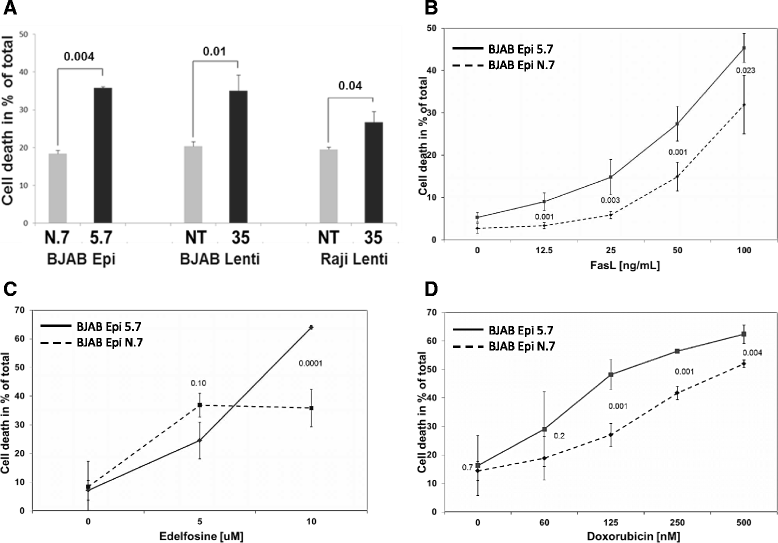
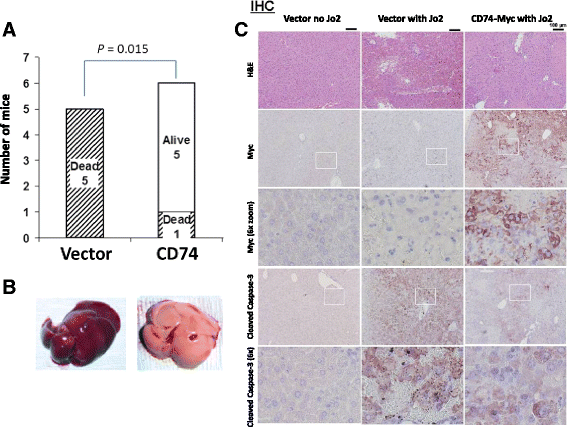
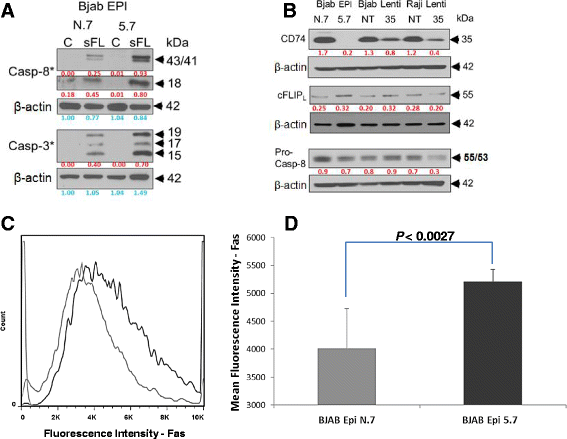
Methods: CD74 expression was suppressed in human B-lymphoma cell lines, BJAB and Raji, by either transduction with lentivirus particles or transfection with episomal vector, both encoding CD74-specific shRNAs or non-target shRNA. Effect of CD74 expression on Fas signaling was evaluated by comparing survival of mice hydrodynamically transfected with vector encoding full-length CD74 or empty vector. Sensitivity of cells with suppressed CD74 expression to FasL, edelfosine, doxorubicin, and a humanized CD74-specific antibody, milatuzumab, was evaluated by flow cytometry and compared to control cells. Fas signaling in response to FasL stimulation and the expression of Fas signaling components were evaluated by Western blot. Surface expression of Fas was detected by flow cytometry.
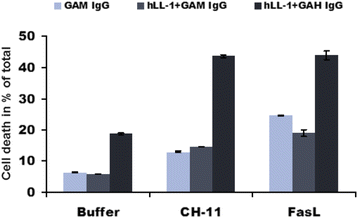
Results: We determined that cells with suppressed CD74 are more sensitive to FasL-induced apoptosis and Fas signaling-dependent chemotherapies, edelfosine and doxorubicin, than control CD74-expressing cells. On the other hand, expression of full-length CD74 in livers protected the mice from a lethal challenge with agonistic anti-Fas antibody Jo2. A detailed analysis of Fas signaling in cells lacking CD74 and control cells revealed increased cleavage/activation of pro-caspase-8 and corresponding enhancement of caspase-3 activation in the absence of CD74, suggesting that CD74 affects the immediate early steps in Fas signaling at the plasma membrane. Cells with suppressed CD74 expression showed increased staining of Fas receptor on their surface. Pre-treatment with milatuzumab sensitized BJAB cells to Fas-mediated apoptosis.
Conclusion: We anticipate that specific targeting of the CD74 on the cell surface will sensitize CD74-expressing cancer cells to Fas-mediated apoptosis, and thus will increase effectiveness of chemotherapy regimens for hematological malignancies.
Figures
Similar articles
-
Milatuzumab-conjugated liposomes as targeted dexamethasone carriers for therapeutic delivery in CD74+ B-cell malignancies.Clin Cancer Res. 2013 Jan 15;19(2):347-56. doi: 10.1158/1078-0432.CCR-12-2046. Epub 2012 Dec 3. Clin Cancer Res. 2013. PMID: 23209030 Free PMC article.
-
Milatuzumab-SN-38 conjugates for the treatment of CD74+ cancers.Mol Cancer Ther. 2013 Jun;12(6):968-78. doi: 10.1158/1535-7163.MCT-12-1170. Epub 2013 Feb 20. Mol Cancer Ther. 2013. PMID: 23427296
-
Nucleolin inhibits Fas ligand binding and suppresses Fas-mediated apoptosis in vivo via a surface nucleolin-Fas complex.Blood. 2013 Jun 6;121(23):4729-39. doi: 10.1182/blood-2012-12-471094. Epub 2013 Apr 18. Blood. 2013. PMID: 23599269 Free PMC article.
-
Milatuzumab - a promising new immunotherapeutic agent.Expert Opin Investig Drugs. 2010 Jan;19(1):141-9. doi: 10.1517/13543780903463854. Expert Opin Investig Drugs. 2010. PMID: 19968579 Review.
-
CD74: a new candidate target for the immunotherapy of B-cell neoplasms.Clin Cancer Res. 2007 Sep 15;13(18 Pt 2):5556s-5563s. doi: 10.1158/1078-0432.CCR-07-1167. Clin Cancer Res. 2007. PMID: 17875789 Review.
Cited by
-
The biological function and significance of CD74 in immune diseases.Inflamm Res. 2017 Mar;66(3):209-216. doi: 10.1007/s00011-016-0995-1. Epub 2016 Oct 17. Inflamm Res. 2017. PMID: 27752708 Review.
-
Knockdown of CD-74 in the Proliferative and Apoptotic Activity of Breast Cancer Cells.Open Access Maced J Med Sci. 2019 Oct 14;7(19):3169-3176. doi: 10.3889/oamjms.2019.354. eCollection 2019 Oct 15. Open Access Maced J Med Sci. 2019. PMID: 31949511 Free PMC article.
-
CD74 in Kidney Disease.Front Immunol. 2015 Sep 23;6:483. doi: 10.3389/fimmu.2015.00483. eCollection 2015. Front Immunol. 2015. PMID: 26441987 Free PMC article. Review.
-
Role of the Macrophage Migration Inhibitory Factor (MIF) in the survival of first trimester human placenta under induced stress conditions.Sci Rep. 2018 Aug 14;8(1):12150. doi: 10.1038/s41598-018-29797-6. Sci Rep. 2018. PMID: 30108299 Free PMC article.
-
T cell help in the autoreactive germinal center.Scand J Immunol. 2022 Jun;95(6):e13192. doi: 10.1111/sji.13192. Epub 2022 May 31. Scand J Immunol. 2022. PMID: 35587582 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

