In vitro expansion of human gastric epithelial stem cells and their responses to bacterial infection
- PMID: 25307862
- PMCID: PMC4274199
- DOI: 10.1053/j.gastro.2014.09.042
In vitro expansion of human gastric epithelial stem cells and their responses to bacterial infection
Abstract
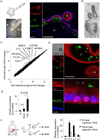
Background & aims: We previously established long-term, 3-dimensional culture of organoids from mouse tissues (intestine, stomach, pancreas, and liver) and human intestine and pancreas. Here we describe conditions required for long-term 3-dimensional culture of human gastric stem cells. The technology can be applied to study the epithelial response to infection with Helicobacter pylori.
Methods: We generated organoids from surgical samples of human gastric corpus. Culture conditions were developed based on those for the mouse gastric and human intestinal systems. We used microinjection to infect the organoids with H pylori. Epithelial responses were measured using microarray and quantitative polymerase chain reaction analyses.
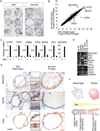
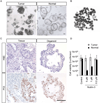
Results: Human gastric cells were expanded indefinitely in 3-dimensional cultures. We cultured cells from healthy gastric tissues, single-sorted stem cells, or tumor tissues. Organoids maintained many characteristics of their respective tissues based on their histology, expression of markers, and euploidy. Organoids from healthy tissue expressed markers of 4 lineages of the stomach and self-organized into gland and pit domains. They could be directed to specifically express either lineages of the gastric gland, or the gastric pit, by addition of nicotinamide and withdrawal of WNT. Although gastric pit lineages had only marginal reactions to bacterial infection, gastric gland lineages mounted a strong inflammatory response.
Conclusions: We developed a system to culture human gastric organoids. This system can be used to study H pylori infection and other gastric pathologies.
Keywords: Gastric Epithelium; Primary Cells; Stomach Cancer; Tissue Engineering.
Copyright © 2015 AGA Institute. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures



References
-
- Karam SM, Leblond CP. Dynamics of epithelial cells in the corpus of the mouse stomach. I. Identification of proliferative cell types and pinpointing of the stem cell. Anat. Rec. 1993;236:259–279. - PubMed
-
- Bjerknes M, Cheng H. Multipotential stem cells in adult mouse gastric epithelium. Am. J. Physiol.- Gastrointest. Liver Physiol. 2002;283:G767–G777. - PubMed
-
- McDonald SAC, Greaves LC, Gutierrez-Gonzalez L, et al. Mechanisms of Field Cancerization in the Human Stomach: The Expansion and Spread of Mutated Gastric Stem Cells. Gastroenterology. 2008;134:500–510. - PubMed
-
- Barker N, Huch M, Kujala P, et al. Lgr5+ve Stem Cells Drive Self-Renewal in the Stomach and Build Long-Lived Gastric Units In Vitro. Cell Stem Cell. 2010;6:25–36. - PubMed
-
- Snippert HJ, Flier LG van der, Sato T, et al. Intestinal Crypt Homeostasis Results from Neutral Competition between Symmetrically Dividing Lgr5 Stem Cells. Cell. 2010;143:134–144. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

