Single cell gel electrophoresis (SCGE) and Pig-a mutation assay in vivo-tools for genotoxicity testing from a regulatory perspective: a study of benzo[a]pyrene in Ogg1(-/-) mice
- PMID: 25308545
- PMCID: PMC4196198
- DOI: 10.1016/j.mrgentox.2014.07.010
Single cell gel electrophoresis (SCGE) and Pig-a mutation assay in vivo-tools for genotoxicity testing from a regulatory perspective: a study of benzo[a]pyrene in Ogg1(-/-) mice
Abstract
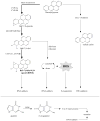
The OECD has developed test guidelines (TG) to identify agents with genotoxic effects. The in vivo alkaline single cell gel electrophoresis (SCGE) assay is currently being prepared to become such a TG. The performance of a combined SCGE/Pig-a gene mutation study was evaluated with the prototypical genotoxicant benzo[a]pyrene (BaP) at an exposure level known to induce germ cell mutation. We aimed to better understand (i) the strengths and weaknesses of the two methods applied in blood and their potential to predict germ cell mutagenicity, and (ii) the involvement of reactive oxygen species (ROS) following in vivo BaP-exposure. To explore the involvement of ROS on BaP genotoxicity, we utilised a mouse model deficient in a DNA glycosylase. Specifically, C57BL/6 mice (Ogg1(+/+) and Ogg1(-/-)) were treated for three consecutive days with 50 mg BaP/kg/day. DNA damage in nucleated blood cells was measured four hours after the last treatment with the SCGE assay, with and without formamidopyrimidine DNA glycosylase (Fpg). Pig-a mutant phenotype blood erythrocytes were analysed two and four weeks after treatment. BaP-induced DNA lesions were not significantly increased in either version of the SCGE assay. The phenotypic mutation frequencies for immature and mature erythrocytes were significantly increased after two weeks. These effects were not affected by genotype, suggesting oxidative damage may have a minor role in BaP genotoxicity, at least in the acute exposure situation studied here. While both assays are promising tools for risk assessment, these results highlight the necessity of understanding the limitations regarding each assay's ability to detect chemicals' genotoxic potential.
Keywords: Alkaline single cell gel electrophoresis (SCGE); Benzo[a]pyrene (BaP); Genotoxicity; Hazard identification; Pig-a mutation assay; Risk assessment.
Copyright © 2014 Elsevier B.V. All rights reserved.
Figures


References
-
- Kirkland D, Speit G. Evaluation of the ability of a battery of three in vitro genotoxicity tests to discriminate rodent carcinogens and non-carcinogens III. Appropriate follow-up testing in vivo. Mutat Res. 2008;654:114–132. - PubMed
-
- Singh NP, McCoy MT, Tice RR, Schneider EL. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res. 1988;175:184–191. - PubMed
-
- Boiteux S, Gajewski E, Laval J, Dizdaroglu M. Substrate specificity of the Escherichia coli Fpg protein (formamidopyrimidine-DNA glycosylase): excision of purine lesions in DNA produced by ionizing radiation or photosensitization. Biochemistry. 1992;31:106–110. - PubMed
-
- Dobrovolsky VN, Miura D, Heflich RH, Dertinger SD. The in vivo Pig-a gene mutation assay, a potential tool for regulatory safety assessment. Environmental and molecular mutagenesis. 2010;51:825–835. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

