Senescence and apoptosis: dueling or complementary cell fates?
- PMID: 25312810
- PMCID: PMC4253488
- DOI: 10.15252/embr.201439245
Senescence and apoptosis: dueling or complementary cell fates?
Abstract
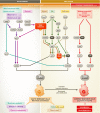
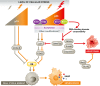
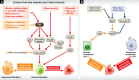
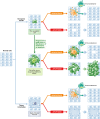
In response to a variety of stresses, mammalian cells undergo a persistent proliferative arrest known as cellular senescence. Many senescence-inducing stressors are potentially oncogenic, strengthening the notion that senescence evolved alongside apoptosis to suppress tumorigenesis. In contrast to apoptosis, senescent cells are stably viable and have the potential to influence neighboring cells through secreted soluble factors, which are collectively known as the senescence-associated secretory phenotype (SASP). However, the SASP has been associated with structural and functional tissue and organ deterioration and may even have tumor-promoting effects, raising the interesting evolutionary question of why apoptosis failed to outcompete senescence as a superior cell fate option. Here, we discuss the advantages that the senescence program may have over apoptosis as a tumor protective mechanism, as well as non-neoplastic functions that may have contributed to its evolution. We also review emerging evidence for the idea that senescent cells are present transiently early in life and are largely beneficial for development, regeneration and homeostasis, and only in advanced age do senescent cells accumulate to an organism's detriment.
Keywords: aging; apoptosis; cancer; embryogenesis; senescence.
© 2014 The Authors.
Figures
Similar articles
-
Pro- and anti-tumorigenic functions of the senescence-associated secretory phenotype.Expert Opin Ther Targets. 2019 Dec;23(12):1041-1051. doi: 10.1080/14728222.2019.1565658. Epub 2019 Jan 15. Expert Opin Ther Targets. 2019. PMID: 30616404 Free PMC article. Review.
-
Impact of senescence-associated secretory phenotype and its potential as a therapeutic target for senescence-associated diseases.Cancer Sci. 2017 Apr;108(4):563-569. doi: 10.1111/cas.13184. Epub 2017 Apr 18. Cancer Sci. 2017. PMID: 28165648 Free PMC article. Review.
-
Senescent cells in cancer therapy: why and how to remove them.Cancer Lett. 2021 Nov 1;520:68-79. doi: 10.1016/j.canlet.2021.07.002. Epub 2021 Jul 5. Cancer Lett. 2021. PMID: 34237406 Review.
-
Keeping the senescence secretome under control: Molecular reins on the senescence-associated secretory phenotype.Exp Gerontol. 2016 Sep;82:39-49. doi: 10.1016/j.exger.2016.05.010. Epub 2016 May 25. Exp Gerontol. 2016. PMID: 27235851 Review.
-
The bright and dark side of extracellular vesicles in the senescence-associated secretory phenotype.Mech Ageing Dev. 2020 Jul;189:111263. doi: 10.1016/j.mad.2020.111263. Epub 2020 May 24. Mech Ageing Dev. 2020. PMID: 32461143 Free PMC article. Review.
Cited by
-
HBP1-mediated Regulation of p21 Protein through the Mdm2/p53 and TCF4/EZH2 Pathways and Its Impact on Cell Senescence and Tumorigenesis.J Biol Chem. 2016 Jun 10;291(24):12688-12705. doi: 10.1074/jbc.M116.714147. Epub 2016 Apr 21. J Biol Chem. 2016. PMID: 27129219 Free PMC article.
-
Ataxia Telangiectasia Presenting as Cervical Dystonia.Cureus. 2022 Oct 26;14(10):e30723. doi: 10.7759/cureus.30723. eCollection 2022 Oct. Cureus. 2022. PMID: 36439585 Free PMC article.
-
Evaluating causality of cellular senescence in non-alcoholic fatty liver disease.JHEP Rep. 2021 May 1;3(4):100301. doi: 10.1016/j.jhepr.2021.100301. eCollection 2021 Aug. JHEP Rep. 2021. PMID: 34113839 Free PMC article. Review.
-
Spindle and Kinetochore-associated Family Genes are Prognostic and Predictive Biomarkers in Hepatocellular Carcinoma.J Clin Transl Hepatol. 2022 Aug 28;10(4):627-641. doi: 10.14218/JCTH.2021.00216. Epub 2022 Jan 4. J Clin Transl Hepatol. 2022. PMID: 36062274 Free PMC article.
-
Single-cell transcriptomic Atlas of aging macaque ocular outflow tissues.Protein Cell. 2024 Jul 20;15(8):594-611. doi: 10.1093/procel/pwad067. Protein Cell. 2024. PMID: 38366188 Free PMC article.
References
-
- Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp Cell Res. 1961;25:585–621. - PubMed
-
- Bodnar AG, Ouellette M, Frolkis M, Holt SE, Chiu CP, Morin GB, Harley CB, Shay JW, Lichtsteiner S, Wright WE, et al. Extension of life-span by introduction of telomerase into normal human cells. Science. 1998;279:349–352. - PubMed
-
- Martinez P, Blasco MA. Telomeric and extra-telomeric roles for telomerase and the telomere-binding proteins. Nat Rev Cancer. 2011;11:161–176. - PubMed
-
- Ouellette MM, McDaniel LD, Wright WE, Shay JW, Schultz RA. The establishment of telomerase-immortalized cell lines representing human chromosome instability syndromes. Hum Mol Genet. 2000;9:403–411. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

