Mucosal immunization with the live attenuated vaccine SPY1 induces humoral and Th2-Th17-regulatory T cell cellular immunity and protects against pneumococcal infection
- PMID: 25312946
- PMCID: PMC4288905
- DOI: 10.1128/IAI.02334-14
Mucosal immunization with the live attenuated vaccine SPY1 induces humoral and Th2-Th17-regulatory T cell cellular immunity and protects against pneumococcal infection
Abstract
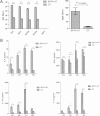
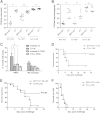
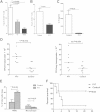
Mucosal immunization with attenuated vaccine can protect against pneumococcal invasion infection, but the mechanism was unknown. Our study found that mucosal delivery with the live attenuated SPY1 vaccine strain can confer T cell- and B cell-dependent protection against pneumococcal colonization and invasive infection; yet it is still unclear which cell subsets contribute to the protection, and their roles in pneumococcal colonization and invasion remain elusive. Adoptive transfer of anti-SPY1 antibody conferred protection to naive μMT mice, and immune T cells were indispensable to protection examined in nude mice. A critical role of interleukin 17A (IL-17A) in colonization was demonstrated in mice lacking IL-17A, and a vaccine-specific Th2 immune subset was necessary for systemic protection. Of note, we found that SPY1 could stimulate an immunoregulatory response and that SPY1-elicited regulatory T cells participated in protection against colonization and lethal infection. The data presented here aid our understanding of how live attenuated strains are able to function as effective vaccines and may contribute to a more comprehensive evaluation of live vaccines and other mucosal vaccines.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures



Similar articles
-
Protective Regulatory T Cell Immune Response Induced by Intranasal Immunization With the Live-Attenuated Pneumococcal Vaccine SPY1 via the Transforming Growth Factor-β1-Smad2/3 Pathway.Front Immunol. 2018 Aug 2;9:1754. doi: 10.3389/fimmu.2018.01754. eCollection 2018. Front Immunol. 2018. PMID: 30116243 Free PMC article.
-
Mucosal and systemic immunization with a novel attenuated pneumococcal vaccine candidate confer serotype independent protection against Streptococcus pneumoniae in mice.Vaccine. 2014 Jul 16;32(33):4179-88. doi: 10.1016/j.vaccine.2014.05.019. Epub 2014 Jun 2. Vaccine. 2014. PMID: 24945468
-
Serotype-independent protection against pneumococcal infections elicited by intranasal immunization with ethanol-killed pneumococcal strain, SPY1.J Microbiol. 2014 Apr;52(4):315-23. doi: 10.1007/s12275-014-3583-5. Epub 2014 Mar 29. J Microbiol. 2014. PMID: 24682994
-
Understanding host immune responses to pneumococcal proteins in the upper respiratory tract to develop serotype-independent pneumococcal vaccines.Expert Rev Vaccines. 2020 Oct;19(10):959-972. doi: 10.1080/14760584.2020.1843433. Epub 2020 Nov 8. Expert Rev Vaccines. 2020. PMID: 33107359 Review.
-
Pneumococcal vaccines: mechanism of action, impact on epidemiology and adaption of the species.Int J Antimicrob Agents. 2008 Sep;32(3):199-206. doi: 10.1016/j.ijantimicag.2008.01.021. Epub 2008 Apr 18. Int J Antimicrob Agents. 2008. PMID: 18378430 Review.
Cited by
-
Combatting Salmonella: a focus on antimicrobial resistance and the need for effective vaccination.BMC Infect Dis. 2025 Jan 20;25(1):84. doi: 10.1186/s12879-025-10478-5. BMC Infect Dis. 2025. PMID: 39833704 Free PMC article.
-
A molecular mucosal adjuvant to enhance immunity against pneumococcal infection in the elderly.Immune Netw. 2015 Feb;15(1):9-15. doi: 10.4110/in.2015.15.1.9. Epub 2015 Feb 17. Immune Netw. 2015. PMID: 25713504 Free PMC article. Review.
-
Streptococcus pneumoniae's Virulence and Host Immunity: Aging, Diagnostics, and Prevention.Front Immunol. 2018 Jun 22;9:1366. doi: 10.3389/fimmu.2018.01366. eCollection 2018. Front Immunol. 2018. PMID: 29988379 Free PMC article. Review.
-
Protective Regulatory T Cell Immune Response Induced by Intranasal Immunization With the Live-Attenuated Pneumococcal Vaccine SPY1 via the Transforming Growth Factor-β1-Smad2/3 Pathway.Front Immunol. 2018 Aug 2;9:1754. doi: 10.3389/fimmu.2018.01754. eCollection 2018. Front Immunol. 2018. PMID: 30116243 Free PMC article.
-
Serotype-Independent Protection Against Invasive Pneumococcal Infections Conferred by Live Vaccine With lgt Deletion.Front Immunol. 2019 May 29;10:1212. doi: 10.3389/fimmu.2019.01212. eCollection 2019. Front Immunol. 2019. PMID: 31191555 Free PMC article.
References
-
- WHO. 2012. Pneumococcal vaccines WHO position paper-2012. Wkly Epidemiol Rec 87:129–144. - PubMed
-
- Flasche S, Van Hoek AJ, Sheasby E, Waight P, Andrews N, Sheppard C, George R, Miller E. 2011. Effect of pneumococcal conjugate vaccination on serotype-specific carriage and invasive disease in England: a cross-sectional study. PLoS Med 8:e1001017. doi:10.1371/journal.pmed.1001017. - DOI - PMC - PubMed
-
- Wu K, Zhang X, Shi J, Li N, Li D, Luo M, Cao J, Yin N, Wang H, Xu W, He Y, Yin Y. 2010. Immunization with a combination of three pneumococcal proteins confers additive and broad protection against Streptococcus pneumoniae infections in mice. Infect Immun 78:1276–1283. doi:10.1128/IAI.00473-09. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

