Antibiotic-loaded synthetic calcium sulfate beads for prevention of bacterial colonization and biofilm formation in periprosthetic infections
- PMID: 25313221
- PMCID: PMC4291338
- DOI: 10.1128/AAC.03676-14
Antibiotic-loaded synthetic calcium sulfate beads for prevention of bacterial colonization and biofilm formation in periprosthetic infections
Abstract
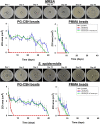
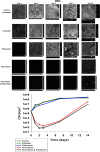
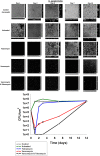
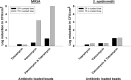
Periprosthetic infection (PI) causes significant morbidity and mortality after fixation and joint arthroplasty and has been extensively linked to the formation of bacterial biofilms. Poly(methyl methacrylate) (PMMA), as a cement or as beads, is commonly used for antibiotic release to the site of infection but displays variable elution kinetics and also represents a potential nidus for infection, therefore requiring surgical removal once antibiotics have eluted. Absorbable cements have shown improved elution of a wider range of antibiotics and, crucially, complete biodegradation, but limited data exist as to their antimicrobial and antibiofilm efficacy. Synthetic calcium sulfate beads loaded with tobramycin, vancomycin, or vancomycin-tobramycin dual treatment (in a 1:0.24 [wt/wt] ratio) were assessed for their abilities to eradicate planktonic methicillin-resistant Staphylococcus aureus (MRSA) and Staphylococcus epidermidis relative to that of PMMA beads. The ability of the calcium sulfate beads to prevent biofilm formation over multiple days and to eradicate preformed biofilms was studied using a combination of viable cell counts, confocal microscopy, and scanning electron microscopy of the bead surface. Biofilm bacteria displayed a greater tolerance to the antibiotics than their planktonic counterparts. Antibiotic-loaded beads were able to kill planktonic cultures of 10(6) CFU/ml, prevent bacterial colonization, and significantly reduce biofilm formation over multiple days. However, established biofilms were harder to eradicate. These data further demonstrate the difficulty in clearing established biofilms; therefore, early preventive measures are key to reducing the risk of PI. Synthetic calcium sulfate loaded with antibiotics has the potential to reduce or eliminate biofilm formation on adjacent periprosthetic tissue and prosthesis material and, thus, to reduce the rates of periprosthetic infection.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures



References
-
- Masterson EL, Masri BA, Duncan CP. 1998. Treatment of infection at the site of total hip replacement. Instr Course Lect 47:297–306. - PubMed
-
- Sculco TP. 1993. The economic impact of infected total joint arthroplasty. Instr Course Lect 42:349–351. - PubMed
-
- Stoodley P, Kathju S, Hu FZ, Erdos G, Levenson JE, Mehta N, Dice B, Johnson S, Hall-Stoodley L, Nistico L, Sotereanos N, Sewecke J, Post JC, Ehrlich GD. 2005. Molecular and imaging techniques for bacterial biofilms in joint arthroplasty infections. Clin Orthop Relat Res 2005:31–40. doi:10.1097/01.blo.0000175129.83084.d5. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

