A role for versican in the development of leiomyosarcoma
- PMID: 25320080
- PMCID: PMC4256343
- DOI: 10.1074/jbc.M114.607168
A role for versican in the development of leiomyosarcoma
Abstract
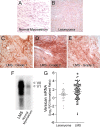
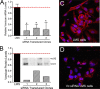
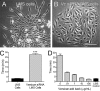
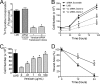
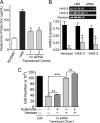
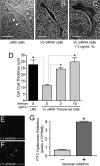
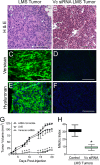
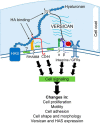
Leiomyosarcoma (LMS) is a mesenchymal cancer that occurs throughout the body. Although LMS is easily recognized histopathologically, the cause of the disease remains unknown. Versican, an extracellular matrix proteoglycan, increases in LMS. Microarray analyses of 80 LMSs and 24 leiomyomas showed a significant elevated expression of versican in human LMS versus benign leiomyomas. To explore the importance of versican in this smooth muscle cell tumor, we used versican-directed siRNA to knock down versican expression in a LMS human cell line, SK-LMS-1. Decreased versican expression was accompanied by slower rates of LMS cell proliferation and migration, increased adhesion, and decreased accumulation of the extracellular matrix macromolecule hyaluronan. Addition of purified versican to cells expressing versican siRNA restored cell proliferation to the level of LMS controls, increased the pericellular coat and the retention of hyaluronan, and decreased cell adhesion in a dose-dependent manner. The presence of versican was not only synergistic with hyaluronan in increasing cell proliferation, but the depletion of versican decreased hyaluronan synthase expression and decreased the retention of hyaluronan. When LMS cells stably expressing versican siRNA were injected into nude mice, the resulting tumors displayed significantly less versican and hyaluronan staining, had lower volumes, and had reduced levels of mitosis as compared with controls. Collectively, these results suggest a role for using versican as a point of control in the management and treatment of LMS.
Keywords: Cell Migration; Cell Proliferation; Extracellular Matrix; Hyaluronan; Leiomyosarcoma; Small Interfering RNA (siRNA); Tumor Growth; Versican.
© 2014 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures
Similar articles
-
Provisional matrix: A role for versican and hyaluronan.Matrix Biol. 2017 Jul;60-61:38-56. doi: 10.1016/j.matbio.2016.12.001. Epub 2016 Dec 6. Matrix Biol. 2017. PMID: 27932299 Free PMC article. Review.
-
Inhibition of versican expression by siRNA facilitates tropoelastin synthesis and elastic fiber formation by human SK-LMS-1 leiomyosarcoma smooth muscle cells in vitro and in vivo.Matrix Biol. 2016 Mar;50:67-81. doi: 10.1016/j.matbio.2015.12.010. Epub 2015 Dec 23. Matrix Biol. 2016. PMID: 26723257 Free PMC article.
-
Platelet-derived growth factor stimulates the formation of versican-hyaluronan aggregates and pericellular matrix expansion in arterial smooth muscle cells.Arch Biochem Biophys. 2001 Oct 1;394(1):29-38. doi: 10.1006/abbi.2001.2507. Arch Biochem Biophys. 2001. PMID: 11566024
-
Estradiol protects dermal hyaluronan/versican matrix during photoaging by release of epidermal growth factor from keratinocytes.J Biol Chem. 2012 Jun 8;287(24):20056-69. doi: 10.1074/jbc.M112.353151. Epub 2012 Apr 9. J Biol Chem. 2012. PMID: 22493503 Free PMC article.
-
Role of versican, hyaluronan and CD44 in ovarian cancer metastasis.Int J Mol Sci. 2011 Jan 31;12(2):1009-29. doi: 10.3390/ijms12021009. Int J Mol Sci. 2011. PMID: 21541039 Free PMC article. Review.
Cited by
-
Expression of V3 Versican by Rat Arterial Smooth Muscle Cells Promotes Differentiated and Anti-inflammatory Phenotypes.J Biol Chem. 2015 Aug 28;290(35):21629-41. doi: 10.1074/jbc.M115.657486. Epub 2015 Jul 7. J Biol Chem. 2015. PMID: 26152723 Free PMC article.
-
The biochemistry and immunohistochemistry of versican.Methods Cell Biol. 2018;143:261-279. doi: 10.1016/bs.mcb.2017.08.015. Epub 2017 Nov 26. Methods Cell Biol. 2018. PMID: 29310782 Free PMC article.
-
V3: an enigmatic isoform of the proteoglycan versican.Am J Physiol Cell Physiol. 2023 Aug 1;325(2):C519-C537. doi: 10.1152/ajpcell.00059.2023. Epub 2023 Jul 3. Am J Physiol Cell Physiol. 2023. PMID: 37399500 Free PMC article. Review.
-
Versican-A Critical Extracellular Matrix Regulator of Immunity and Inflammation.Front Immunol. 2020 Mar 24;11:512. doi: 10.3389/fimmu.2020.00512. eCollection 2020. Front Immunol. 2020. PMID: 32265939 Free PMC article. Review.
-
Provisional matrix: A role for versican and hyaluronan.Matrix Biol. 2017 Jul;60-61:38-56. doi: 10.1016/j.matbio.2016.12.001. Epub 2016 Dec 6. Matrix Biol. 2017. PMID: 27932299 Free PMC article. Review.
References
-
- Major F. J., Blessing J. A., Silverberg S. G., Morrow C. P., Creasman W. T., Currie J. L., Yordan E., Brady M. F. (1993) Prognostic factors in early-stage uterine sarcoma. A Gynecologic Oncology Group study. Cancer 71, 1702–1709 - PubMed
-
- Moinfar F., Azodi M., Tavassoli F. A. (2007) Uterine sarcomas. Pathology 39, 55–71 - PubMed
-
- Zako M., Shinomura T., Ujita M., Ito K., Kimata K. (1995) Expression of PG-M (V3), an alternatively spliced form of PG-M without a chondroitin sulfate attachment region in mouse and human tissues. J. Biol. Chem. 270, 3914–3918 - PubMed
-
- Lemire J. M., Braun K. R., Maurel P., Kaplan E. D., Schwartz S. M., Wight T. N. (1999) Versican/PG-M isoforms in vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 19, 1630–1639 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

