Resistance to antifungals that target CYP51
- PMID: 25320648
- PMCID: PMC4182338
- DOI: 10.1007/s12154-014-0121-1
Resistance to antifungals that target CYP51
Abstract
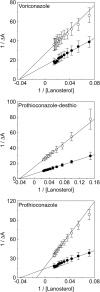
Fungal diseases are an increasing global burden. Fungi are now recognised to kill more people annually than malaria, whilst in agriculture, fungi threaten crop yields and food security. Azole resistance, mediated by several mechanisms including point mutations in the target enzyme (CYP51), is increasing through selection pressure as a result of widespread use of triazole fungicides in agriculture and triazole antifungal drugs in the clinic. Mutations similar to those seen in clinical isolates as long ago as the 1990s in Candida albicans and later in Aspergillus fumigatus have been identified in agriculturally important fungal species and also wider combinations of point mutations. Recently, evidence that mutations originate in the field and now appear in clinical infections has been suggested. This situation is likely to increase in prevalence as triazole fungicide use continues to rise. Here, we review the progress made in understanding azole resistance found amongst clinically and agriculturally important fungal species focussing on resistance mechanisms associated with CYP51. Biochemical characterisation of wild-type and mutant CYP51 enzymes through ligand binding studies and azole IC50 determinations is an important tool for understanding azole susceptibility and can be used in conjunction with microbiological methods (MIC50 values), molecular biological studies (site-directed mutagenesis) and protein modelling studies to inform future antifungal development with increased specificity for the target enzyme over the host homologue.
Keywords: Antifungals; Azole resistance; CYP51; Fungicides; Point mutations; Sterol 14-demethylase.
Figures







References
-
- Shelton BK. Opportunistic fungal infections in the critically ill. Crit Care Nurs Clin N Am. 2000;12(3):323–340. - PubMed
-
- GAFFI (2014) Global action fund for fungal infection; http://www.gaffi.org/
-
- Havlickova B, Czaika VA, Friedrich M. Epidemiological trends in skin mycoses worldwide. Mycoses. 2008;51(Suppl 4):2–15. - PubMed
-
- Matee MI, Scheutz F, Moshy J. Occurrence of oral lesions in relation to clinical and immunological status among HIV-infected adult Tanzanians. Oral Dis. 2000;6(2):106–111. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

