Sensory gating of an embryonic zebrafish interneuron during spontaneous motor behaviors
- PMID: 25324729
- PMCID: PMC4179717
- DOI: 10.3389/fncir.2014.00121
Sensory gating of an embryonic zebrafish interneuron during spontaneous motor behaviors
Abstract
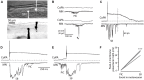
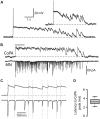
In all but the simplest monosynaptic reflex arcs, sensory stimuli are encoded by sensory neurons that transmit a signal via sensory interneurons to downstream partners in order to elicit a response. In the embryonic zebrafish (Danio rerio), cutaneous Rohon-Beard (RB) sensory neurons fire in response to mechanical stimuli and excite downstream glutamatergic commissural primary ascending (CoPA) interneurons to produce a flexion response contralateral to the site of stimulus. In the absence of sensory stimuli, zebrafish spinal locomotor circuits are spontaneously active during development due to pacemaker activity resulting in repetitive coiling of the trunk. Self-generated movement must therefore be distinguishable from external stimuli in order to ensure the appropriate activation of touch reflexes. Here, we recorded from CoPAs during spontaneous and evoked fictive motor behaviors in order to examine how responses to self-movement are gated in sensory interneurons. During spontaneous coiling, CoPAs received glycinergic inputs coincident with contralateral flexions that shunted firing for the duration of the coiling event. Shunting inactivation of CoPAs was caused by a slowly deactivating chloride conductance that resulted in lowered membrane resistance and increased action potential threshold. During spontaneous burst swimming, which develops later, CoPAs received glycinergic inputs that arrived in phase with excitation to ipsilateral motoneurons and provided persistent shunting. During a touch stimulus, short latency glutamatergic inputs produced cationic currents through AMPA receptors that drove a single, large amplitude action potential in the CoPA before shunting inhibition began, providing a brief window for the activation of downstream neurons. We compared the properties of CoPAs to those of other spinal neurons and propose that glycinergic signaling onto CoPAs acts as a corollary discharge signal for reflex inhibition during movement.
Keywords: AMPA receptors; corollary discharge; glycine receptors; reflex inhibition; sensory interneurons; spinal cord; spontaneous behavior; zebrafish.
Figures




Similar articles
-
A hybrid electrical/chemical circuit in the spinal cord generates a transient embryonic motor behavior.J Neurosci. 2014 Jul 16;34(29):9644-55. doi: 10.1523/JNEUROSCI.1225-14.2014. J Neurosci. 2014. PMID: 25031404 Free PMC article.
-
Glutamate drives the touch response through a rostral loop in the spinal cord of zebrafish embryos.Dev Neurobiol. 2009 Oct;69(12):780-95. doi: 10.1002/dneu.20741. Dev Neurobiol. 2009. PMID: 19634126 Free PMC article.
-
ALX 1393 inhibits spontaneous network activity by inducing glycinergic tonic currents in the spinal ventral horn.Neuroscience. 2013 Dec 3;253:165-71. doi: 10.1016/j.neuroscience.2013.08.042. Epub 2013 Aug 29. Neuroscience. 2013. PMID: 23994185
-
Spinal cords: Symphonies of interneurons across species.Front Neural Circuits. 2023 Apr 26;17:1146449. doi: 10.3389/fncir.2023.1146449. eCollection 2023. Front Neural Circuits. 2023. PMID: 37180760 Free PMC article. Review.
-
Measured motion: searching for simplicity in spinal locomotor networks.Curr Opin Neurobiol. 2009 Dec;19(6):572-86. doi: 10.1016/j.conb.2009.10.011. Epub 2009 Nov 10. Curr Opin Neurobiol. 2009. PMID: 19896834 Free PMC article. Review.
Cited by
-
Unmyelinated sensory neurons use Neuregulin signals to promote myelination of interneurons in the CNS.Cell Rep. 2022 Nov 15;41(7):111669. doi: 10.1016/j.celrep.2022.111669. Cell Rep. 2022. PMID: 36384112 Free PMC article.
-
Position- and quantity-dependent responses in zebrafish turning behavior.Sci Rep. 2016 Jun 13;6:27888. doi: 10.1038/srep27888. Sci Rep. 2016. PMID: 27292818 Free PMC article.
-
Single-Cell Reconstruction of Emerging Population Activity in an Entire Developing Circuit.Cell. 2019 Oct 3;179(2):355-372.e23. doi: 10.1016/j.cell.2019.08.039. Epub 2019 Sep 26. Cell. 2019. PMID: 31564455 Free PMC article.
-
Behavioral screening of the LOPAC1280 library in zebrafish embryos.Toxicol Appl Pharmacol. 2017 Aug 15;329:241-248. doi: 10.1016/j.taap.2017.06.011. Epub 2017 Jun 13. Toxicol Appl Pharmacol. 2017. PMID: 28623180 Free PMC article.
-
Spinal V1 neurons inhibit motor targets locally and sensory targets distally.Curr Biol. 2021 Sep 13;31(17):3820-3833.e4. doi: 10.1016/j.cub.2021.06.053. Epub 2021 Jul 21. Curr Biol. 2021. PMID: 34289387 Free PMC article.
References
-
- Ali D. W., Buss R. R., Drapeau P. (2000a). Properties of miniature glutamatergic EPSCs in neurons of the locomotor regions of the developing zebrafish. J. Neurophysiol. 83 181–191 - PubMed
-
- Ali D. W., Drapeau P., Legendre P. (2000b). Development of spontaneous glycinergic currents in the Mauthner neuron of the zebrafish embryo. J. Neurophysiol. 84 1726–1736 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

