A transgenic approach to live imaging of heparan sulfate modification patterns
- PMID: 25325959
- PMCID: PMC5893304
- DOI: 10.1007/978-1-4939-1714-3_22
A transgenic approach to live imaging of heparan sulfate modification patterns
Abstract
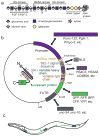
Heparan sulfate (HS) glycosaminoglycan chains contain highly modified HS domains that are separated by sections of sparse or no modification. HS domains are central to the role of HS in protein binding and mediating protein-protein interactions in the extracellular matrix. Since HS domains are not genetically encoded, they are impossible to visualize and study with conventional methods in vivo. Here we describe a transgenic approach using previously described single chain variable fragment (scFv) antibodies that bind HS in vitro and on tissue sections with different specificities. By engineering a secretion signal and a fluorescent protein to the scFvs and transgenically expressing these fluorescently tagged antibodies in Caenorhabditis elegans, we are able to directly visualize specific HS domains in live animals (Attreed et al. Nat Methods 9(5):477-479, 2012). The approach allows concomitant colabeling of multiple epitopes, the study of HS dynamics and, could lend itself to a genetic analysis of HS domain biosynthesis or to visualize other nongenetically encoded or posttranslational modifications.
Figures

References
-
- Bernfield M, Götte M, Park PW, Reizes O, Fitzgerald ML, Lincecum J, Zako M. Functions of cell surface heparan sulfate proteoglycans. Annu Rev Biochem. 1999;68:729–777. - PubMed
-
- Bishop JR, Schuksz M, Esko JD. Heparan sulphate proteoglycans fine-tune mammalian physiology. Nature. 2007;446:1030–1037. - PubMed
-
- Bülow HE, Hobert O. The Molecular Diversity of Glycosaminoglycans Shapes Animal Development. Ann Rev Cell Dev Biol. 2006;22:375–407. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

