Protein kinase IKKβ-catalyzed phosphorylation of IRF5 at Ser462 induces its dimerization and nuclear translocation in myeloid cells
- PMID: 25326418
- PMCID: PMC4267347
- DOI: 10.1073/pnas.1418399111
Protein kinase IKKβ-catalyzed phosphorylation of IRF5 at Ser462 induces its dimerization and nuclear translocation in myeloid cells
Abstract
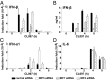
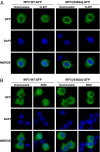
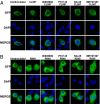
The siRNA knockdown of IFN Regulatory Factor 5 (IRF5) in the human plasmacytoid dendritic cell line Gen2.2 prevented IFNβ production induced by compound CL097, a ligand for Toll-like receptor 7 (TLR7). CL097 also stimulated the phosphorylation of IRF5 at Ser462 and stimulated the nuclear translocation of wild-type IRF5, but not the IRF5[Ser462Ala] mutant. The CL097-stimulated phosphorylation of IRF5 at Ser462 and its nuclear translocation was prevented by the pharmacological inhibition of protein kinase IKKβ or the siRNA knockdown of IKKβ or its "upstream" activator, the protein kinase TAK1. Similar results were obtained in a murine macrophage cell line stimulated with the TLR7 agonist compound R848 or the nucleotide oligomerization domain 1 (NOD1) agonist KF-1B. IKKβ phosphorylated IRF5 at Ser462 in vitro and induced the dimerization of wild-type IRF5 but not the IRF5[S462A] mutant. These findings demonstrate that IKKβ activates two "master" transcription factors of the innate immune system, IRF5 and NF-κB.
Keywords: IKKβ; IRF5; TLR7; interferon β; plasmacytoid dendritic cell.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Comment in
-
Innate sense of purpose for IKKβ.Proc Natl Acad Sci U S A. 2014 Dec 9;111(49):17348-9. doi: 10.1073/pnas.1419689111. Epub 2014 Nov 26. Proc Natl Acad Sci U S A. 2014. PMID: 25427798 Free PMC article. No abstract available.
References
-
- Takaoka A, et al. Integral role of IRF-5 in the gene induction programme activated by Toll-like receptors. Nature. 2005;434(7030):243–249. - PubMed
-
- Yasuda K, et al. Murine dendritic cell type I IFN production induced by human IgG-RNA immune complexes is IFN regulatory factor (IRF)5 and IRF7 dependent and is required for IL-6 production. J Immunol. 2007;178(11):6876–6885. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

