IKKβ is an IRF5 kinase that instigates inflammation
- PMID: 25326420
- PMCID: PMC4267374
- DOI: 10.1073/pnas.1418516111
IKKβ is an IRF5 kinase that instigates inflammation
Abstract
The transcription factor interferon regulatory factor 5 (IRF5) is essential for the induction of inflammatory cytokines, but the mechanism by which IRF5 is activated is not well understood. Here we present evidence that the kinase IKKβ phosphorylates and activates IRF5 in response to stimulation in several inflammatory pathways, including those emanated from Toll-like receptors and retinoic acid-inducible gene I-like receptors. IKKβ phosphorylates mouse IRF5 at specific residues, including serine 445 (S446 in human IRF5 isoform 1), as evidenced by mass spectrometry analysis and detection with a phosphospecific antibody. Recombinant IKKβ phosphorylated IRF5 at Ser-445 in vitro, and a point mutation of this serine abolished IRF5 activation and cytokine production. Depletion or pharmacologic inhibition of IKKβ prevented IRF5 phosphorylation. These results indicate that IKKβ is an IRF5 kinase that instigates inflammation.
Keywords: IKK; IRF5; TLR; inflammation; phosphorylation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
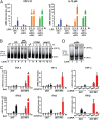
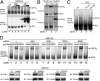

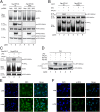
Comment in
-
Innate sense of purpose for IKKβ.Proc Natl Acad Sci U S A. 2014 Dec 9;111(49):17348-9. doi: 10.1073/pnas.1419689111. Epub 2014 Nov 26. Proc Natl Acad Sci U S A. 2014. PMID: 25427798 Free PMC article. No abstract available.
References
-
- Honda K, Taniguchi T. IRFs: Master regulators of signalling by Toll-like receptors and cytosolic pattern-recognition receptors. Nat Rev Immunol. 2006;6(9):644–658. - PubMed
-
- Tamura T, Yanai H, Savitsky D, Taniguchi T. The IRF family transcription factors in immunity and oncogenesis. Annu Rev Immunol. 2008;26:535–584. - PubMed
-
- Ikushima H, Negishi H, Taniguchi T. The IRF family transcription factors at the interface of innate and adaptive immune responses. Cold Spring Harb Symp Quant Biol. 2013;78:105–116. - PubMed
-
- Kawai T, et al. IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nat Immunol. 2005;6(10):981–988. - PubMed
-
- Meylan E, et al. Cardif is an adaptor protein in the RIG-I antiviral pathway and is targeted by hepatitis C virus. Nature. 2005;437(7062):1167–1172. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

