The SIRT1 activator SRT1720 reverses vascular endothelial dysfunction, excessive superoxide production, and inflammation with aging in mice
- PMID: 25326534
- PMCID: PMC4269699
- DOI: 10.1152/ajpheart.00377.2014
The SIRT1 activator SRT1720 reverses vascular endothelial dysfunction, excessive superoxide production, and inflammation with aging in mice
Abstract
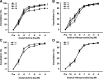
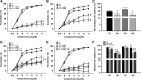
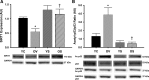
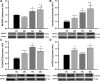
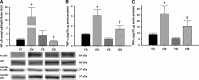
Reductions in arterial SIRT1 expression and activity with aging are linked to vascular endothelial dysfunction. We tested the hypothesis that the specific SIRT1 activator SRT1720 improves endothelial function [endothelium-dependent dilation (EDD)] in old mice. Young (4-9 mo) and old (29-32 mo) male B6D2F1 mice treated with SRT1720 (100 mg/kg body wt) or vehicle for 4 wk were studied with a group of young controls. Compared with the young controls, aortic SIRT1 expression and activity were reduced (P < 0.05) and EDD was impaired (83 ± 2 vs. 96 ± 1%; P < 0.01) in old vehicle-treated animals. SRT1720 normalized SIRT1 expression/activity in old mice and restored EDD (95 ± 1%) by enhancing cyclooxygenase (COX)-2-mediated dilation and protein expression in the absence of changes in nitric oxide bioavailability. Aortic superoxide production and expression of NADPH oxidase 4 (NOX4) were increased in old vehicle mice (P < 0.05), and ex vivo administration of the superoxide scavenger TEMPOL restored EDD in that group. SRT1720 normalized aortic superoxide production in old mice, without altering NOX4 and abolished the improvement in EDD with TEMPOL, while selectively increasing aortic antioxidant enzymes. Aortic nuclear factor-κB (NF-κB) activity and tumor necrosis factor-α (TNF-α) were increased in old vehicle mice (P < 0.05), whereas SRT1720 normalized NF-κB activation and reduced TNF-α in old animals. SIRT1 activation with SRT1720 ameliorates vascular endothelial dysfunction with aging in mice by enhancing COX-2 signaling and reducing oxidative stress and inflammation. Specific activation of SIRT1 is a promising therapeutic strategy for age-related endothelial dysfunction in humans.
Keywords: COX-2 dilation; SIRT1; aging; superoxide; vascular inflammation.
Figures


Similar articles
-
Nitrite supplementation reverses vascular endothelial dysfunction and large elastic artery stiffness with aging.Aging Cell. 2011 Jun;10(3):429-37. doi: 10.1111/j.1474-9726.2011.00679.x. Epub 2011 Mar 31. Aging Cell. 2011. PMID: 21276184 Free PMC article.
-
Apigenin restores endothelial function by ameliorating oxidative stress, reverses aortic stiffening, and mitigates vascular inflammation with aging.Am J Physiol Heart Circ Physiol. 2021 Jul 1;321(1):H185-H196. doi: 10.1152/ajpheart.00118.2021. Epub 2021 Jun 11. Am J Physiol Heart Circ Physiol. 2021. PMID: 34114892 Free PMC article.
-
Voluntary wheel running restores endothelial function in conduit arteries of old mice: direct evidence for reduced oxidative stress, increased superoxide dismutase activity and down-regulation of NADPH oxidase.J Physiol. 2009 Jul 1;587(Pt 13):3271-85. doi: 10.1113/jphysiol.2009.169771. Epub 2009 May 5. J Physiol. 2009. PMID: 19417091 Free PMC article.
-
Antagonistic crosstalk between NF-κB and SIRT1 in the regulation of inflammation and metabolic disorders.Cell Signal. 2013 Oct;25(10):1939-48. doi: 10.1016/j.cellsig.2013.06.007. Epub 2013 Jun 11. Cell Signal. 2013. PMID: 23770291 Review.
-
Endothelial SIRT1 as a Target for the Prevention of Arterial Aging: Promises and Challenges.J Cardiovasc Pharmacol. 2021 Dec 1;78(Suppl 6):S63-S77. doi: 10.1097/FJC.0000000000001154. J Cardiovasc Pharmacol. 2021. PMID: 34840264 Review.
Cited by
-
Mitochondrial contributions to vascular endothelial dysfunction, arterial stiffness, and cardiovascular diseases.Am J Physiol Heart Circ Physiol. 2021 May 1;320(5):H2080-H2100. doi: 10.1152/ajpheart.00917.2020. Epub 2021 Apr 9. Am J Physiol Heart Circ Physiol. 2021. PMID: 33834868 Free PMC article. Review.
-
Treatment with recombinant Sirt1 rewires the cardiac lipidome and rescues diabetes-related metabolic cardiomyopathy.Cardiovasc Diabetol. 2023 Nov 13;22(1):312. doi: 10.1186/s12933-023-02057-2. Cardiovasc Diabetol. 2023. PMID: 37957697 Free PMC article.
-
SIRT1-PGC1α-NFκB Pathway of Oxidative and Inflammatory Stress during Trypanosoma cruzi Infection: Benefits of SIRT1-Targeted Therapy in Improving Heart Function in Chagas Disease.PLoS Pathog. 2016 Oct 20;12(10):e1005954. doi: 10.1371/journal.ppat.1005954. eCollection 2016 Oct. PLoS Pathog. 2016. PMID: 27764247 Free PMC article.
-
Sirtuins and NAD+ in the Development and Treatment of Metabolic and Cardiovascular Diseases.Circ Res. 2018 Sep 14;123(7):868-885. doi: 10.1161/CIRCRESAHA.118.312498. Circ Res. 2018. PMID: 30355082 Free PMC article. Review.
-
Implicating endothelial cell senescence to dysfunction in the ageing and diseased brain.Basic Clin Pharmacol Toxicol. 2020 Aug;127(2):102-110. doi: 10.1111/bcpt.13403. Epub 2020 Mar 23. Basic Clin Pharmacol Toxicol. 2020. PMID: 32162446 Free PMC article. Review.
References
-
- Alcendor RR, Gao S, Zhai P, Zablocki D, Holle E, Yu X, Tian B, Wagner T, Vatner SF, Sadoshima J. Sirt1 regulates aging and resistance to oxidative stress in the heart. Circ Res 100: 1512–1521, 2007. - PubMed
-
- Blackwell KA, Sorenson JP, Richardson DM, Smith LA, Suda O, Nath K, Katusic ZS. Mechanisms of aging-induced impairment of endothelium-dependent relaxation: role of tetrahydrobiopterin. Am J Physiol Heart Circ Physiol 287: H2448–H2453, 2004. - PubMed
-
- Cawello W, Schweer H, Muller R, Bonn R, Seyberth HW. Metabolism and pharmacokinetics of prostaglandin E1 administered by intravenous infusion in human subjects. Eur J Clin Pharmacol 46: 275–277, 1994. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

