HER2 activation results in β-catenin-dependent changes in pulmonary epithelial permeability
- PMID: 25326580
- PMCID: PMC4338945
- DOI: 10.1152/ajplung.00237.2014
HER2 activation results in β-catenin-dependent changes in pulmonary epithelial permeability
Abstract
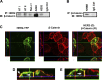
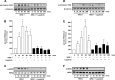
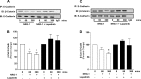
The receptor tyrosine kinase human epidermal growth factor receptor-2 (HER2) is known to regulate pulmonary epithelial barrier function; however, the mechanisms behind this effect remain unidentified. We hypothesized that HER2 signaling alters the epithelial barrier through an interaction with the adherens junction (AJ) protein β-catenin, leading to dissolution of the AJ. In quiescent pulmonary epithelial cells, HER2 and β-catenin colocalized along the lateral intercellular junction. HER2 activation by the ligand neuregulin-1 was associated with tyrosine phosphorylation of β-catenin, dissociation of β-catenin from E-cadherin, and decreased E-cadherin-mediated cell adhesion. All effects were blocked with the HER2 inhibitor lapatinib. β-Catenin knockdown using shRNA significantly attenuated neuregulin-1-induced decreases in pulmonary epithelial resistance in vitro. Our data indicate that HER2 interacts with β-catenin, leading to dissolution of the AJ, decreased cell-cell adhesion, and disruption of the pulmonary epithelial barrier.
Keywords: adherens junction; cell adhesion; epithelial cell; human epidermal growth factor receptor-2; neuregulin-1; permeability; receptor tyrosine kinase; β-catenin.
Copyright © 2015 the American Physiological Society.
Figures



References
-
- Akhmetshina A, Palumbo K, Dees C, Bergmann C, Venalis P, Zerr P, Horn A, Kireva T, Beyer C, Zwerina J, Schneider H, Sadowski A, Riener MO, MacDougald OA, Distler O, Schett G, Distler JH. Activation of canonical Wnt signalling is required for TGF-beta-mediated fibrosis. Nat Commun 3: 735, 2012. - PMC - PubMed
-
- Aumiller V, Balsara N, Wilhelm J, Gunther A, Konigshoff M. WNT/beta-catenin signaling induces IL-1beta expression by alveolar epithelial cells in pulmonary fibrosis. Am J Respir Cell Mol Biol 49: 96–104, 2013. - PubMed
-
- Beckers J, Herrmann F, Rieger S, Drobyshev AL, Horsch M, Hrabé de Angelis M, Seliger B. Identification and validation of novel ERBB2 (HER2, NEU) targets including genes involved in angiogenesis. Int J Cancer 114: 590–597, 2005. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

