Megakaryocytes regulate hematopoietic stem cell quiescence through CXCL4 secretion
- PMID: 25326802
- PMCID: PMC4258871
- DOI: 10.1038/nm.3707
Megakaryocytes regulate hematopoietic stem cell quiescence through CXCL4 secretion
Abstract
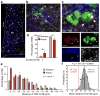
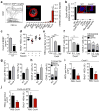
In the bone marrow, hematopoietic stem cells (HSCs) lodge in specialized microenvironments that tightly control the proliferative state of HSCs to adapt to the varying needs for replenishment of blood cells while also preventing HSC exhaustion. All putative niche cells suggested thus far have a nonhematopoietic origin. Thus, it remains unclear how feedback from mature cells is conveyed to HSCs to adjust their proliferation. Here we show that megakaryocytes (MKs) can directly regulate HSC pool size in mice. Three-dimensional whole-mount imaging revealed that endogenous HSCs are frequently located adjacent to MKs in a nonrandom fashion. Selective in vivo depletion of MKs resulted in specific loss of HSC quiescence and led to a marked expansion of functional HSCs. Gene expression analyses revealed that MKs are the source of chemokine C-X-C motif ligand 4 (CXCL4, also named platelet factor 4 or PF4) in the bone marrow, and we found that CXCL4 regulates HSC cell cycle activity. CXCL4 injection into mice resulted in a reduced number of HSCs because of their increased quiescence. By contrast, Cxcl4(-/-) mice exhibited an increased number of HSCs and increased HSC proliferation. Combined use of whole-mount imaging and computational modeling was highly suggestive of a megakaryocytic niche capable of independently influencing HSC maintenance by regulating quiescence. These results indicate that a terminally differentiated cell type derived from HSCs contributes to the HSC niche, directly regulating HSC behavior.
Figures


References
-
- Orford KW, Scadden DT. Deconstructing stem cell self-renewal: genetic insights into cell-cycle regulation. Nat Rev Genet. 2008;9:115–128. - PubMed
-
- Bianco P. Bone and the hematopoietic niche: a tale of two stem cells. Blood. 2011;117:5281–5288. - PubMed
-
- Yamazaki S, et al. Nonmyelinating schwann cells maintain hematopoietic stem cell hibernation in the bone marrow niche. Cell. 2011;147:1146–1158. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- U01 HL099656/HL/NHLBI NIH HHS/United States
- T32 063754/PHS HHS/United States
- DA033788/DA/NIDA NIH HHS/United States
- R01 HL069438/HL/NHLBI NIH HHS/United States
- T32 GM062754/GM/NIGMS NIH HHS/United States
- P01 HL064190/HL/NHLBI NIH HHS/United States
- P01 HL110860/HL/NHLBI NIH HHS/United States
- R01 CA164468/CA/NCI NIH HHS/United States
- R01 DA033788/DA/NIDA NIH HHS/United States
- CA164468/CA/NCI NIH HHS/United States
- U01 HL099993/HL/NHLBI NIH HHS/United States
- P30 CA013330/CA/NCI NIH HHS/United States
- R01 DK056638/DK/NIDDK NIH HHS/United States
- R01 HL069438/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

