Specific microbiome changes in a mouse model of parenteral nutrition associated liver injury and intestinal inflammation
- PMID: 25329595
- PMCID: PMC4203793
- DOI: 10.1371/journal.pone.0110396
Specific microbiome changes in a mouse model of parenteral nutrition associated liver injury and intestinal inflammation
Abstract
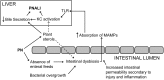
Background: Parenteral nutrition (PN) has been a life-saving treatment in infants intolerant of enteral feedings. However, PN is associated with liver injury (PN Associated Liver Injury: PNALI) in a significant number of PN-dependent infants. We have previously reported a novel PNALI mouse model in which PN infusion combined with intestinal injury results in liver injury. In this model, lipopolysaccharide activation of toll-like receptor 4 signaling, soy oil-derived plant sterols, and pro-inflammatory activation of Kupffer cells (KCs) played key roles. The objective of this study was to explore changes in the intestinal microbiome associated with PNALI.
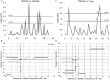
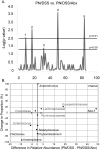
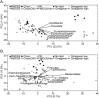
Methodology and principal findings: Microbiome analysis in the PNALI mouse identified specific alterations within colonic microbiota associated with PNALI and further association of these communities with the lipid composition of the PN solution. Intestinal inflammation or soy oil-based PN infusion alone (in the absence of enteral feeds) caused shifts within the gut microbiota. However, the combination resulted in accumulation of a specific taxon, Erysipelotrichaceae (23.8% vs. 1.7% in saline infused controls), in PNALI mice. Moreover, PNALI was markedly attenuated by enteral antibiotic treatment, which also was associated with significant reduction of Erysipelotrichaceae (0.6%) and a Gram-negative constituent, the S24-7 lineage of Bacteroidetes (53.5% in PNALI vs. 0.8%). Importantly, removal of soy oil based-lipid emulsion from the PN solution resulted in significant reduction of Erysipelotrichaceae as well as attenuation of PNALI. Finally, addition of soy-derived plant sterol (stigmasterol) to fish oil-based PN restored Erysipelotrichaceae abundance and PNALI.
Conclusions: Soy oil-derived plant sterols and the associated specific bacterial groups in the colonic microbiota are associated with PNALI. Products from these bacteria may directly trigger activation of KCs and promote PNALI. Furthermore, the results indicate that lipid modification of PN solutions may alter specific intestinal bacterial species associated with PNALI, and thus suggest strategies for management of PNALI.
Conflict of interest statement
Figures

Similar articles
-
Toll-like receptor 4-dependent Kupffer cell activation and liver injury in a novel mouse model of parenteral nutrition and intestinal injury.Hepatology. 2012 May;55(5):1518-28. doi: 10.1002/hep.25500. Epub 2012 Mar 18. Hepatology. 2012. PMID: 22120983 Free PMC article.
-
Phytosterols promote liver injury and Kupffer cell activation in parenteral nutrition-associated liver disease.Sci Transl Med. 2013 Oct 9;5(206):206ra137. doi: 10.1126/scitranslmed.3006898. Sci Transl Med. 2013. PMID: 24107776 Free PMC article.
-
Preserved Gut Microbial Diversity Accompanies Upregulation of TGR5 and Hepatobiliary Transporters in Bile Acid-Treated Animals Receiving Parenteral Nutrition.JPEN J Parenter Enteral Nutr. 2017 Feb;41(2):198-207. doi: 10.1177/0148607116661838. Epub 2016 Aug 20. JPEN J Parenter Enteral Nutr. 2017. PMID: 27503935 Free PMC article.
-
Changes to the Intestinal Microbiome With Parenteral Nutrition: Review of a Murine Model and Potential Clinical Implications.Nutr Clin Pract. 2015 Dec;30(6):798-806. doi: 10.1177/0884533615609904. Epub 2015 Sep 30. Nutr Clin Pract. 2015. PMID: 26424591 Review.
-
Parenteral Nutrition-Associated Liver Disease: The Role of the Gut Microbiota.Nutrients. 2017 Sep 7;9(9):987. doi: 10.3390/nu9090987. Nutrients. 2017. PMID: 28880224 Free PMC article. Review.
Cited by
-
Increasing Evidence That Irritable Bowel Syndrome and Functional Gastrointestinal Disorders Have a Microbial Pathogenesis.Front Cell Infect Microbiol. 2020 Sep 9;10:468. doi: 10.3389/fcimb.2020.00468. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 33014892 Free PMC article. Review.
-
Change in the Gut Microbiota of Lactating Sows and Their Piglets by Inclusion of Dietary Spray-Dried Plasma in Sow Diets.J Microbiol Biotechnol. 2024 Mar 28;34(3):516-524. doi: 10.4014/jmb.2311.11001. Epub 2023 Dec 15. J Microbiol Biotechnol. 2024. PMID: 38111306 Free PMC article.
-
Metabolic and Microbiome Alterations Following the Enrichment of a High-Fat Diet With High Oleic Acid Peanuts Versus the Traditional Peanuts Cultivar in Mice.Front Nutr. 2022 Jun 15;9:823756. doi: 10.3389/fnut.2022.823756. eCollection 2022. Front Nutr. 2022. PMID: 35782916 Free PMC article.
-
Burn Injury Leads to Increase in Relative Abundance of Opportunistic Pathogens in the Rat Gastrointestinal Microbiome.Front Microbiol. 2017 Jul 6;8:1237. doi: 10.3389/fmicb.2017.01237. eCollection 2017. Front Microbiol. 2017. PMID: 28729860 Free PMC article.
-
Intestinal Microbiota, Lipids, and the Pathogenesis of Intestinal Failure-Associated Liver Disease.J Pediatr. 2015 Sep;167(3):519-26. doi: 10.1016/j.jpeds.2015.05.048. Epub 2015 Jun 27. J Pediatr. 2015. PMID: 26130113 Free PMC article. No abstract available.
References
-
- Wilmore DW, Dudrick SJ (1968) Growth and development of an infant receiving all nutrients exclusively by vein. JAMA 203: 860–864. - PubMed
-
- Jeejeebhoy KN (2001) Total parenteral nutrition: potion or poison? Am J Clin Nutr 74: 160–163. - PubMed
-
- Kelly DA (2006) Intestinal failure-associated liver disease: what do we know today? Gastroenterology 130: S70–77. - PubMed
-
- Sokol RJ (1997) Total parenteral nutrition-related liver disease. Zhonghua Min Guo Xiao Er Ke Yi Xue Hui Za Zhi 38: 418–428. - PubMed
-
- Teitelbaum DH (1997) Parenteral nutrition-associated cholestasis. Curr Opin Pediatr 9: 270–275. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

