HIV-1 subtype C unproductively infects human cardiomyocytes in vitro and induces apoptosis mitigated by an anti-Gp120 aptamer
- PMID: 25329893
- PMCID: PMC4201581
- DOI: 10.1371/journal.pone.0110930
HIV-1 subtype C unproductively infects human cardiomyocytes in vitro and induces apoptosis mitigated by an anti-Gp120 aptamer
Abstract
HIV-associated cardiomyopathy (HIVCM) is of clinical concern in developing countries because of a high HIV-1 prevalence, especially subtype C, and limited access to highly active antiretroviral therapy (HAART). For these reasons, we investigated the direct and indirect effects of HIV-1 subtype C infection of cultured human cardiomyocytes and the mechanisms leading to cardiomyocytes damage; as well as a way to mitigate the damage. We evaluated a novel approach to mitigate HIVCM using a previously reported gp120 binding and HIV-1 neutralizing aptamer called UCLA1. We established a cell-based model of HIVCM by infecting human cardiomyocytes with cell-free HIV-1 or co-culturing human cardiomyocytes with HIV-infected monocyte derived macrophages (MDM). We discovered that HIV-1 subtype C unproductively (i.e. its life cycle is arrested after reverse transcription) infects cardiomyocytes. Furthermore, we found that HIV-1 initiates apoptosis of cardiomyocytes through caspase-9 activation, preferentially via the intrinsic or mitochondrial initiated pathway. CXCR4 receptor-using viruses were stronger inducers of apoptosis than CCR5 utilizing variants. Importantly, we discovered that HIV-1 induced apoptosis of cardiomyocytes was mitigated by UCLA1. However, UCLA1 had no protective effective on cardiomyocytes when apoptosis was triggered by HIV-infected MDM. When HIV-1 was treated with UCLA1 prior to infection of MDM, it failed to induce apoptosis of cardiomyocytes. These data suggest that HIV-1 causes a mitochondrial initiated apoptotic cascade, which signal through caspase-9, whereas HIV-1 infected MDM causes apoptosis predominantly via the death-receptor pathway, mediated by caspase-8. Furthermore the data suggest that UCLA1 protects cardiomyocytes from caspase-mediated apoptosis, directly by binding to HIV-1 and indirectly by preventing infection of MDM.
Conflict of interest statement
Figures
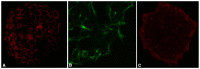
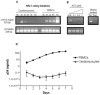
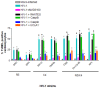
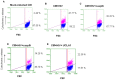
Similar articles
-
HIV-1 induces cardiomyopathyby cardiomyocyte invasion and gp120, Tat, and cytokine apoptotic signaling.Cardiovasc Toxicol. 2004;4(2):97-107. doi: 10.1385/ct:4:2:097. Cardiovasc Toxicol. 2004. PMID: 15371627
-
Role of HIV-1 subtype C envelope V3 to V5 regions in viral entry, coreceptor utilization and replication efficiency in primary T-lymphocytes and monocyte-derived macrophages.Virol J. 2007 Nov 24;4:126. doi: 10.1186/1743-422X-4-126. Virol J. 2007. PMID: 18036244 Free PMC article.
-
Cardiomyocytes undergo apoptosis in human immunodeficiency virus cardiomyopathy through mitochondrion- and death receptor-controlled pathways.Proc Natl Acad Sci U S A. 2002 Oct 29;99(22):14386-91. doi: 10.1073/pnas.212327899. Epub 2002 Oct 11. Proc Natl Acad Sci U S A. 2002. PMID: 12379743 Free PMC article.
-
HIV-1 subtype C predicted co-receptor tropism in Africa: an individual sequence level meta-analysis.AIDS Res Ther. 2020 Feb 7;17(1):5. doi: 10.1186/s12981-020-0263-x. AIDS Res Ther. 2020. PMID: 32033571 Free PMC article. Review.
-
HIV-1 envelope, integrins and co-receptor use in mucosal transmission of HIV.J Transl Med. 2011 Jan 27;9 Suppl 1(Suppl 1):S2. doi: 10.1186/1479-5876-9-S1-S2. J Transl Med. 2011. PMID: 21284901 Free PMC article. Review.
Cited by
-
Human immunodeficiency virus infection and risks of morbidity and death in adults with incident heart failure.Eur Heart J Open. 2021 Dec 1;1(3):oeab040. doi: 10.1093/ehjopen/oeab040. eCollection 2021 Nov. Eur Heart J Open. 2021. PMID: 35919879 Free PMC article.
-
Oligonucleotide aptamers: promising and powerful diagnostic and therapeutic tools for infectious diseases.J Infect. 2018 Aug;77(2):83-98. doi: 10.1016/j.jinf.2018.04.007. Epub 2018 May 7. J Infect. 2018. PMID: 29746951 Free PMC article. Review.
-
Declining Incidence of Systolic Left Ventricular Dysfunction in Human Immunodeficiency Virus-Infected Individuals Treated With Highly Active Antiretroviral Therapy.Am J Cardiol. 2016 Apr 1;117(7):1194-5. doi: 10.1016/j.amjcard.2016.01.008. Epub 2016 Jan 14. Am J Cardiol. 2016. PMID: 26993978 Free PMC article. No abstract available.
-
HIV, highly active antiretroviral therapy and the heart: a cellular to epidemiological review.HIV Med. 2016 Jun;17(6):411-24. doi: 10.1111/hiv.12346. Epub 2015 Nov 26. HIV Med. 2016. PMID: 26611380 Free PMC article. Review.
-
Diversity and Inter-Connections in the CXCR4 Chemokine Receptor/Ligand Family: Molecular Perspectives.Front Immunol. 2015 Aug 21;6:429. doi: 10.3389/fimmu.2015.00429. eCollection 2015. Front Immunol. 2015. PMID: 26347749 Free PMC article. Review.
References
-
- Ntsekhe M, Hakim J (2005) Impact of human immunodeficiency virus infection on cardiovascular disease in Africa. Circulation 112: 3602–3607. - PubMed
-
- Ntsekhe M, Mayosi BM (2009) Cardiac manifestations of HIV infection: an African perspective. Nat Clin Pract Cardiovasc Med 6: 120–127. - PubMed
-
- Magula NP, Mayosi BM (2003) Cardiac involvement in HIV-infected people living in Africa: a review. Cardiovasc J S Afr 14: 231–237. - PubMed
-
- Fiala M, Popik W, Qiao JH, Lossinsky AS, Alce T, et al. (2004) HIV-1 induces cardiomyopathyby cardiomyocyte invasion and gp120, Tat, and cytokine apoptotic signaling. Cardiovasc Toxicol 4: 97–107. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

