WWOX, the common fragile site FRA16D gene product, regulates ATM activation and the DNA damage response
- PMID: 25331887
- PMCID: PMC4226089
- DOI: 10.1073/pnas.1409252111
WWOX, the common fragile site FRA16D gene product, regulates ATM activation and the DNA damage response
Abstract
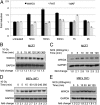
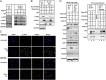
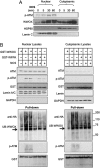
Genomic instability is a hallmark of cancer. The WW domain-containing oxidoreductase (WWOX) is a tumor suppressor spanning the common chromosomal fragile site FRA16D. Here, we report a direct role of WWOX in DNA damage response (DDR) and DNA repair. We show that Wwox deficiency results in reduced activation of the ataxia telangiectasia-mutated (ATM) checkpoint kinase, inefficient induction and maintenance of γ-H2AX foci, and impaired DNA repair. Mechanistically, we show that, upon DNA damage, WWOX accumulates in the cell nucleus, where it interacts with ATM and enhances its activation. Nuclear accumulation of WWOX is regulated by its K63-linked ubiquitination at lysine residue 274, which is mediated by the E3 ubiquitin ligase ITCH. These findings identify a novel role for the tumor suppressor WWOX and show that loss of WWOX expression may drive genomic instability and provide an advantage for clonal expansion of neoplastic cells.
Keywords: ITCH; WW domain-containing oxidoreductase; ataxia telangiectasia-mutated; common fragile sites; genomic instability.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Bartek J, Lukas J. DNA damage checkpoints: From initiation to recovery or adaptation. Curr Opin Cell Biol. 2007;19(2):238–245. - PubMed
-
- Harper JW, Elledge SJ. The DNA damage response: Ten years after. Mol Cell. 2007;28(5):739–745. - PubMed
-
- Shiloh Y. ATM and ATR: Networking cellular responses to DNA damage. Curr Opin Genet Dev. 2001;11(1):71–77. - PubMed
-
- Falck J, Coates J, Jackson SP. Conserved modes of recruitment of ATM, ATR and DNA-PKcs to sites of DNA damage. Nature. 2005;434(7033):605–611. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

