Endochondral ossification for enhancing bone regeneration: converging native extracellular matrix biomaterials and developmental engineering in vivo
- PMID: 25336144
- PMCID: PMC4442558
- DOI: 10.1089/ten.TEB.2014.0419
Endochondral ossification for enhancing bone regeneration: converging native extracellular matrix biomaterials and developmental engineering in vivo
Abstract
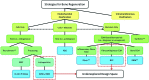
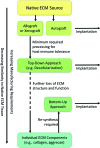
Autologous bone grafting (ABG) remains entrenched as the gold standard of treatment in bone regenerative surgery. Consequently, many marginally successful bone tissue engineering strategies have focused on mimicking portions of ABG's "ideal" osteoconductive, osteoinductive, and osteogenic composition resembling the late reparative stage extracellular matrix (ECM) in bone fracture repair, also known as the "hard" or "bony" callus. An alternative, less common approach that has emerged in the last decade harnesses endochondral (EC) ossification through developmental engineering principles, which acknowledges that the molecular and cellular mechanisms involved in developmental skeletogenesis, specifically EC ossification, are closely paralleled during native bone healing. EC ossification naturally occurs during the majority of bone fractures and, thus, can potentially be utilized to enhance bone regeneration for nearly any orthopedic indication, especially in avascular critical-sized defects where hypoxic conditions favor initial chondrogenesis instead of direct intramembranous ossification. The body's native EC ossification response, however, is not capable of regenerating critical-sized defects without intervention. We propose that an underexplored potential exists to regenerate bone through the native EC ossification response by utilizing strategies which mimic the initial inflammatory or fibrocartilaginous ECM (i.e., "pro-" or "soft" callus) observed in the early reparative stage of bone fracture repair. To date, the majority of strategies utilizing this approach rely on clinically burdensome in vitro cell expansion protocols. This review will focus on the confluence of two evolving areas, (1) native ECM biomaterials and (2) developmental engineering, which will attempt to overcome the technical, business, and regulatory challenges that persist in the area of bone regeneration. Significant attention will be given to native "raw" materials and ECM-based designs that provide necessary osteo- and chondro-conductive and inductive features for enhancing EC ossification. In addition, critical perspectives on existing stem cell-based therapeutic strategies will be discussed with a focus on their use as an extension of the acellular ECM-based designs for specific clinical indications. Within this framework, a novel realm of unexplored design strategies for bone tissue engineering will be introduced into the collective consciousness of the regenerative medicine field.
Figures

Similar articles
-
Recapitulating endochondral ossification: a promising route to in vivo bone regeneration.J Tissue Eng Regen Med. 2015 Aug;9(8):889-902. doi: 10.1002/term.1918. Epub 2014 Jun 11. J Tissue Eng Regen Med. 2015. PMID: 24916192 Review.
-
Fractionated human adipose tissue as a native biomaterial for the generation of a bone organ by endochondral ossification.Acta Biomater. 2018 Sep 1;77:142-154. doi: 10.1016/j.actbio.2018.07.004. Epub 2018 Jul 4. Acta Biomater. 2018. PMID: 30126590
-
The cell and molecular biology of fracture healing.Clin Orthop Relat Res. 1998 Oct;(355 Suppl):S7-21. doi: 10.1097/00003086-199810001-00003. Clin Orthop Relat Res. 1998. PMID: 9917622
-
Histological evolution of the regenerate during bone transport: an experimental study in sheep.Injury. 2016 Sep;47 Suppl 3:S7-S14. doi: 10.1016/S0020-1383(16)30600-3. Injury. 2016. PMID: 27692111
-
Biology of Bone Formation, Fracture Healing, and Distraction Osteogenesis.J Craniofac Surg. 2017 Jul;28(5):1380-1389. doi: 10.1097/SCS.0000000000003625. J Craniofac Surg. 2017. PMID: 28562424 Review.
Cited by
-
Exploring calcium-free alternatives in endochondral bone repair tested on In vivo trials - A review.Regen Ther. 2024 Jun 1;26:145-160. doi: 10.1016/j.reth.2024.05.017. eCollection 2024 Jun. Regen Ther. 2024. PMID: 38872977 Free PMC article. Review.
-
Multifunctional coatings of nickel-titanium implant toward promote osseointegration after operation of bone tumor and clinical application: a review.Front Bioeng Biotechnol. 2024 Feb 20;12:1325707. doi: 10.3389/fbioe.2024.1325707. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 38444648 Free PMC article. Review.
-
Bone remodeling effect of a chitosan and calcium phosphate-based composite.Regen Biomater. 2019 Aug;6(4):241-247. doi: 10.1093/rb/rbz009. Epub 2019 Mar 18. Regen Biomater. 2019. PMID: 31402983 Free PMC article.
-
Evaluation of an Engineered Hybrid Matrix for Bone Regeneration via Endochondral Ossification.Ann Biomed Eng. 2020 Mar;48(3):992-1005. doi: 10.1007/s10439-019-02279-0. Epub 2019 Apr 29. Ann Biomed Eng. 2020. PMID: 31037444 Free PMC article.
-
Nanocrystalline hydroxyapatite-poly(thioketal urethane) nanocomposites stimulate a combined intramembranous and endochondral ossification response in rabbits.ACS Biomater Sci Eng. 2020 Jan 13;6(1):564-574. doi: 10.1021/acsbiomaterials.9b01378. Epub 2019 Dec 10. ACS Biomater Sci Eng. 2020. PMID: 32405537 Free PMC article.
References
-
- Einhorn T.A. The science of fracture healing. J Orthop Trauma 19, S4, 2005 - PubMed
-
- Gerstenfeld L.C., Cullinane D.M., Barnes G.L., Graves D.T., and Einhorn T.A. Fracture healing as a post-natal developmental process: molecular, spatial, and temporal aspects of its regulation. J Cell Biochem 88, 873, 2003 - PubMed
-
- Kalfas I.H. Principles of bone healing. Neurosurg Focus 10, E1, 2001 - PubMed
-
- Meyer U., and Weismann H.P. Bone and cartilage. In: Schroeder G., ed. Bone and Cartilage Engineering. New York, NY: Springer, 2006, pp. 7–46
-
- Miclau T., Schneider R.A., Eames B.F., and Helms J.A. Common molecular mechanisms regulating fetal bone formation and adult fracture repair. In: Lieberman J.R., and Friedlaender G.E., eds. Bone Regeneration and Repair: Biology and Clinical Application. Totowa, NJ: Humana Press, Inc., 2005, pp. 45–55
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

