Ca(2+) signaling in arterioles and small arteries of conscious, restrained, optical biosensor mice
- PMID: 25339912
- PMCID: PMC4188025
- DOI: 10.3389/fphys.2014.00387
Ca(2+) signaling in arterioles and small arteries of conscious, restrained, optical biosensor mice
Abstract
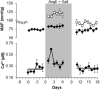
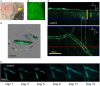
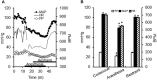
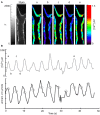
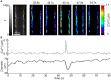
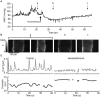
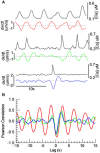
Two-photon fluorescence microscopy and conscious, restrained optical biosensor mice were used to study smooth muscle Ca(2+) signaling in ear arterioles. Conscious mice were used in order to preserve normal mean arterial blood pressure (MAP) and sympathetic nerve activity (SNA). ExMLCK mice, which express a genetically-encoded smooth muscle-specific FRET-based Ca(2+) indicator, were equipped with blood pressure telemetry and immobilized for imaging. MAP was 101 ± 4 mmHg in conscious restrained mice, similar to the freely mobile state (107 ± 3 mmHg). Oscillatory vasomotion or irregular contractions were observed in most arterioles (71%), with the greatest oscillatory frequency observed at 0.25 s(-1). In a typical arteriole with an average diameter of ~35 μm, oscillatory vasomotion of a 5-6 μm magnitude was accompanied by nearly uniform [Ca(2+)] oscillations from ~0.1 to 0.5 μM, with maximum [Ca(2+)] occurring immediately before the rapid decrease in diameter. Very rapid, spatially uniform "Ca(2+) flashes" were also observed but not asynchronous propagating Ca(2+) waves. In contrast, vasomotion and dynamic Ca(2+) signals were rarely observed in ear arterioles of anesthetized exMLCK biosensor mice. Hexamethonium (30 μg/g BW, i.p.) caused a fall in MAP to 74 ± 4 mmHg, arteriolar vasodilation, and abolition of vasomotion and synchronous Ca(2+) transients.
Summary: MAP and heart rate (HR) were normal during high-resolution Ca(2+) imaging of conscious, restrained mice. SNA induced continuous vasomotion and irregular vasoconstrictions via spatially uniform Ca(2+) signaling within the arterial wall. FRET-based biosensor mice and two-photon imaging provided the first measurements of [Ca(2+)] in vascular smooth muscle cells in arterioles of conscious animals.
Keywords: Calcium; arteriole; imaging; sympathetic nervous system; two-photon.
Figures
Similar articles
-
Imaging sympathetic neurogenic Ca2+ signaling in blood vessels.Auton Neurosci. 2017 Nov;207:59-66. doi: 10.1016/j.autneu.2017.07.007. Epub 2017 Jul 29. Auton Neurosci. 2017. PMID: 28781164 Free PMC article. Review.
-
A method for noninvasive longitudinal measurements of [Ca2+] in arterioles of hypertensive optical biosensor mice.Am J Physiol Heart Circ Physiol. 2014 Jul 15;307(2):H173-81. doi: 10.1152/ajpheart.00182.2014. Epub 2014 May 23. Am J Physiol Heart Circ Physiol. 2014. PMID: 24858846 Free PMC article.
-
Vascular tone and Ca(2+) signaling in murine cremaster muscle arterioles in vivo.Microcirculation. 2013 Apr;20(3):269-77. doi: 10.1111/micc.12025. Microcirculation. 2013. PMID: 23140521 Free PMC article.
-
Synchrony of spontaneous Ca2+ activity in microvascular mural cells.J Smooth Muscle Res. 2020;56(0):1-18. doi: 10.1540/jsmr.56.1. J Smooth Muscle Res. 2020. PMID: 32249242 Free PMC article. Review.
-
Cellular and Ionic Mechanisms of Arterial Vasomotion.Adv Exp Med Biol. 2019;1124:297-312. doi: 10.1007/978-981-13-5895-1_12. Adv Exp Med Biol. 2019. PMID: 31183832 Review.
Cited by
-
Emptying the Pool: Modular Insulin Secretion From the Pancreas.Diabetes. 2016 Mar;65(3):542-4. doi: 10.2337/dbi15-0041. Diabetes. 2016. PMID: 26908903 Free PMC article. No abstract available.
-
The role of capsaicin-sensitive C-fiber afferent pathways in the control of micturition in spinal-intact and spinal cord-injured mice.Am J Physiol Renal Physiol. 2017 Sep 1;313(3):F796-F804. doi: 10.1152/ajprenal.00097.2017. Epub 2017 Jun 21. Am J Physiol Renal Physiol. 2017. PMID: 28637786 Free PMC article.
-
Smooth Muscle Ion Channels and Regulation of Vascular Tone in Resistance Arteries and Arterioles.Compr Physiol. 2017 Mar 16;7(2):485-581. doi: 10.1002/cphy.c160011. Compr Physiol. 2017. PMID: 28333380 Free PMC article. Review.
-
Imaging sympathetic neurogenic Ca2+ signaling in blood vessels.Auton Neurosci. 2017 Nov;207:59-66. doi: 10.1016/j.autneu.2017.07.007. Epub 2017 Jul 29. Auton Neurosci. 2017. PMID: 28781164 Free PMC article. Review.
-
Venous cerebral blood volume increase during voluntary locomotion reflects cardiovascular changes.Neuroimage. 2015 Sep;118:301-12. doi: 10.1016/j.neuroimage.2015.06.011. Epub 2015 Jun 6. Neuroimage. 2015. PMID: 26057593 Free PMC article.
References
-
- Blaustein M. P., Leenen F. H., Chen L., Golovina V. A., Hamlyn J. M., Pallone T. L., et al. (2012). How NaCl raises blood pressure: a new paradigm for the pathogenesis of salt-dependent hypertension. Am. J. Physiol. Heart Circ. Physiol. 302, H1031–H1049 10.1152/ajpheart.00899.2011 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

