Identification and molecular characterization of a chitin-binding protein from the beet webworm, Loxostege sticticalis L
- PMID: 25340980
- PMCID: PMC4227266
- DOI: 10.3390/ijms151019147
Identification and molecular characterization of a chitin-binding protein from the beet webworm, Loxostege sticticalis L
Abstract
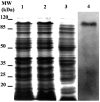
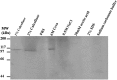
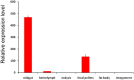
As the first crucial barrier in the midgut of insects, the peritrophic membrane (PM) plays an important role in preventing external invasion. PM proteins, as the major components of the PM, determine the structure and function of this membrane. A new PM protein, named LstiCBP, from the PM of Loxostege sticticalis larvae was identified using cDNA library screening. The full cDNA of LstiCBP is 2606 bp in length and contains a 2403 bp ORF that encodes an 808-amino acid preprotein with a 15-amino acid as signal peptide. The deduced protein sequence of the cDNA contains 8 cysteine-rich chitin-binding domains (CBDs). Recombinant LstiCBP was successfully expressed in BL21 cells using recombinant plasmid DNA and showed high chitin-binding activity. LstiCBP expression was detected in the midgut at both the transcriptional and translational levels; however, the biochemical and physiological functions of LstiCBP in L. sticticalis require further investigation.
Figures



Similar articles
-
Identification and molecular characterization of a new member of the peritrophic membrane proteins from the meadow moth, loxostege sticticalis.Int J Biol Sci. 2010 Sep 6;6(5):491-8. doi: 10.7150/ijbs.6.491. Int J Biol Sci. 2010. PMID: 20827401 Free PMC article.
-
CONSTRUCTION OF SILKWORM MIDGUT cDNA LIBRARY FOR SCREEN AND SEQUENCE ANALYSIS OF PERITROPHIC MEMBRANE PROTEIN GENES.Arch Insect Biochem Physiol. 2016 Jan;91(1):3-16. doi: 10.1002/arch.21305. Epub 2015 Oct 16. Arch Insect Biochem Physiol. 2016. PMID: 26472595
-
Identification of two new peritrophic membrane proteins from larval Trichoplusia ni: structural characteristics and their functions in the protease rich insect gut.Insect Biochem Mol Biol. 2004 Mar;34(3):215-27. doi: 10.1016/j.ibmb.2003.10.001. Insect Biochem Mol Biol. 2004. PMID: 14871618
-
Molecular Characterization of a Lysozyme Gene and Its Altered Expression Profile in Crowded Beet Webworm (Loxostege sticticalis).PLoS One. 2016 Aug 30;11(8):e0161384. doi: 10.1371/journal.pone.0161384. eCollection 2016. PLoS One. 2016. PMID: 27575006 Free PMC article.
-
The peritrophic matrix of hematophagous insects.Arch Insect Biochem Physiol. 2001 Jun;47(2):119-25. doi: 10.1002/arch.1042. Arch Insect Biochem Physiol. 2001. PMID: 11376458 Review.
Cited by
-
Capsicumicine, a New Bioinspired Peptide from Red Peppers Prevents Staphylococcal Biofilm In Vitro and In Vivo via a Matrix Anti-Assembly Mechanism of Action.Microbiol Spectr. 2021 Oct 31;9(2):e0047121. doi: 10.1128/Spectrum.00471-21. Epub 2021 Oct 27. Microbiol Spectr. 2021. PMID: 34704807 Free PMC article.
-
Identification of Peritrophins and Antiviral Effect of Bm01504 against BmNPV in the Silkworm, Bombyx mori.Int J Mol Sci. 2020 Oct 27;21(21):7973. doi: 10.3390/ijms21217973. Int J Mol Sci. 2020. PMID: 33121000 Free PMC article.
-
Identification of miRNAs Involved in Olfactory Regulation in Antennae of Beet Webworm, Loxostege sticticalis (Lepidoptera: Pyralidae).Life (Basel). 2024 Dec 23;14(12):1705. doi: 10.3390/life14121705. Life (Basel). 2024. PMID: 39768411 Free PMC article.
-
A Thermophile-Fermented Compost Modulates Intestinal Cations and the Expression of a Juvenile Hormone-Binding Protein Gene in the Female Larvae of Hercules Beetle Dynastes hercules (Coleoptera: Scarabaeidae).Insects. 2023 Nov 27;14(12):910. doi: 10.3390/insects14120910. Insects. 2023. PMID: 38132584 Free PMC article.
References
-
- Ji H.H., Yuan Z.M. Peritrophic membrane: A potential target for biocontrol of pest insects. Acta Entomol. Sin. 2005;48:968–974.
-
- Casu R., Eisemann C.H., Pearson R.D., Riding G.A., East L.J., Donaldson A., Cadogan L., Tellam R.L. Antibody-mediated inhibition of the growth of larvae from an insect causing sutaneous myiasis in a mammalian host. Proc. Natl. Acad. Sci. USA. 1997;94:8939–8944. doi: 10.1073/pnas.94.17.8939. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

