Inositol synthesis regulates the activation of GSK-3α in neuronal cells
- PMID: 25345501
- PMCID: PMC4393753
- DOI: 10.1111/jnc.12978
Inositol synthesis regulates the activation of GSK-3α in neuronal cells
Abstract
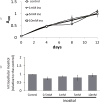
The synthesis of inositol provides precursors of inositol lipids and inositol phosphates that are pivotal for cell signaling. Mood stabilizers lithium and valproic acid, used for treating bipolar disorder, cause cellular inositol depletion, which has been proposed as a therapeutic mechanism of action of both drugs. Despite the importance of inositol, the requirement for inositol synthesis in neuronal cells is not well understood. Here, we examined inositol effects on proliferation of SK-N-SH neuroblastoma cells. The essential role of inositol synthesis in proliferation is underscored by the findings that exogenous inositol was dispensable for proliferation, and inhibition of inositol synthesis decreased proliferation. Interestingly, the inhibition of inositol synthesis by knocking down INO1, which encodes inositol-3-phosphate synthase, the rate-limiting enzyme of inositol synthesis, led to the inactivation of GSK-3α by increasing the inhibitory phosphorylation of this kinase. Similarly, the mood stabilizer valproic acid effected transient decreases in intracellular inositol, leading to inactivation of GSK-3α. As GSK-3 inhibition has been proposed as a likely therapeutic mechanism of action, the finding that inhibition of inositol synthesis results in the inactivation of GSK-3α suggests a unifying hypothesis for mechanism of mood-stabilizing drugs. Inositol is an essential metabolite that serves as a precursor for inositol lipids and inositol phosphates. We report that inhibition of the rate-limiting enzyme of inositol synthesis leads to the inactivation of glycogen synthase kinase (GSK) 3α by increasing inhibitory phosphorylation of this kinase. These findings have implications for the therapeutic mechanisms of mood stabilizers and suggest that inositol synthesis and GSK 3α activity are intrinsically related.
Keywords: Inositol; bipolar disorder; glycogen synthesis kinase; inositol depletion; myo-inositol-3-phosphate synthase; valproic acid.
© 2014 International Society for Neurochemistry.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures



Similar articles
-
The common inositol-reversible effect of mood stabilizers on neurons does not involve GSK3 inhibition, myo-inositol-1-phosphate synthase or the sodium-dependent myo-inositol transporters.Mol Cell Neurosci. 2006 May-Jun;32(1-2):27-36. doi: 10.1016/j.mcn.2006.01.015. Epub 2006 Mar 13. Mol Cell Neurosci. 2006. PMID: 16531065
-
Glycogen synthase kinase-3 is required for optimal de novo synthesis of inositol.Mol Microbiol. 2007 Feb;63(4):1248-58. doi: 10.1111/j.1365-2958.2007.05591.x. Mol Microbiol. 2007. PMID: 17257308
-
Up-regulation of protein L-isoaspartyl methyltransferase expression by lithium is mediated by glycogen synthase kinase-3 inactivation and beta-catenin stabilization.Neuropharmacology. 2008 Oct;55(5):669-76. doi: 10.1016/j.neuropharm.2008.05.033. Epub 2008 Jun 6. Neuropharmacology. 2008. PMID: 18582478
-
Search for a common mechanism of mood stabilizers.Biochem Pharmacol. 2003 Jul 15;66(2):179-89. doi: 10.1016/s0006-2952(03)00187-4. Biochem Pharmacol. 2003. PMID: 12826261 Review.
-
Glycogen synthase kinase-3: a putative molecular target for lithium mimetic drugs.Neuropsychopharmacology. 2005 Jul;30(7):1223-37. doi: 10.1038/sj.npp.1300731. Neuropsychopharmacology. 2005. PMID: 15827567 Review.
Cited by
-
Myo-inositol phosphate synthase expression in the European eel (Anguilla anguilla) and Nile tilapia (Oreochromis niloticus): effect of seawater acclimation.Am J Physiol Regul Integr Comp Physiol. 2016 Aug 1;311(2):R287-98. doi: 10.1152/ajpregu.00056.2016. Epub 2016 Jun 1. Am J Physiol Regul Integr Comp Physiol. 2016. PMID: 27252471 Free PMC article.
-
Inositol Hexakisphosphate Kinase 1 (IP6K1) Regulates Inositol Synthesis in Mammalian Cells.J Biol Chem. 2016 May 13;291(20):10437-44. doi: 10.1074/jbc.M116.714816. Epub 2016 Mar 7. J Biol Chem. 2016. PMID: 26953345 Free PMC article.
-
Disruption of INOS, a Gene Encoding myo-Inositol Phosphate Synthase, Causes Male Sterility in Drosophila melanogaster.G3 (Bethesda). 2018 Aug 30;8(9):2913-2922. doi: 10.1534/g3.118.200403. G3 (Bethesda). 2018. PMID: 29991509 Free PMC article.
-
Inositol Depletion Induced by Acute Treatment of the Bipolar Disorder Drug Valproate Increases Levels of Phytosphingosine.J Biol Chem. 2017 Mar 24;292(12):4953-4959. doi: 10.1074/jbc.M117.775460. Epub 2017 Jan 18. J Biol Chem. 2017. PMID: 28100786 Free PMC article.
-
Inositol depletion regulates phospholipid metabolism and activates stress signaling in HEK293T cells.Biochim Biophys Acta Mol Cell Biol Lipids. 2022 Jun;1867(6):159137. doi: 10.1016/j.bbalip.2022.159137. Epub 2022 Mar 3. Biochim Biophys Acta Mol Cell Biol Lipids. 2022. PMID: 35247568 Free PMC article.
References
-
- Acevedo LD, Holloway HW, Rapoport SI, Shetty HU. Application of stable isotope tracer combined with mass spectrometric detection for studying myo-inositol uptake by cultured neurons from fetal mouse: effect of trisomy 16. J Mass Spectrom. 1997;32:395–400. - PubMed
-
- Allison JH, Stewart MA. Reduced brain inositol in lithium-treated rats. Nature: New biology. 1971;233:267–268. - PubMed
-
- Aukema HM, Holub BJ. Inositol and pyrroloquinoline quinone. A: inositol. Lea and Febiger; Philadelphia: 1994.
-
- Azab AN, He Q, Ju S, Li G, Greenberg ML. Glycogen synthase kinase-3 is required for optimal de novo synthesis of inositol. Mol Microbiol. 2007;63:1248–1258. - PubMed
-
- Bachhawat N, Ouyang Q, Henry SA. Functional characterization of an inositol-sensitive upstream activation sequence in yeast. A cis-regulatory element responsible for inositol-choline mediated regulation of phospholipid biosynthesis. J Biol Chem. 1995;270:25087–25095. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

