Novel Peptidomimetics for Inhibition of HER2:HER3 Heterodimerization in HER2-Positive Breast Cancer
- PMID: 25346057
- PMCID: PMC4408264
- DOI: 10.1111/cbdd.12453
Novel Peptidomimetics for Inhibition of HER2:HER3 Heterodimerization in HER2-Positive Breast Cancer
Abstract
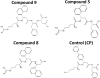
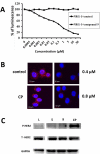
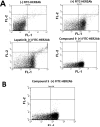
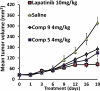
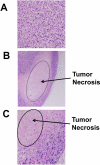
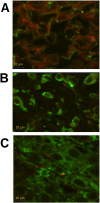
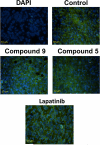
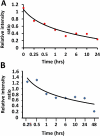
The current approach to treating HER2-overexpressed breast cancer is the use of monoclonal antibodies or a combination of antibodies with traditional chemotherapeutic agents or kinase inhibitors. Our approach is to target clinically validated HER2 domain IV with peptidomimetics and inhibit the protein-protein interactions (PPI) of HERs. Unlike antibodies, peptidomimetics have advantages in terms of stability, modification, and molecular size. We have designed peptidomimetics (compounds 5 and 9) that bind to HER2 domain IV, inhibit protein-protein interactions, and decrease cell viability in breast cancer cells with HER2 overexpression. We have shown, using enzyme fragment complementation and proximity ligation assays, that peptidomimetics inhibit the PPI of HER2:HER3. Compounds 5 and 9 suppressed the tumor growth in a xenograft mouse model. Furthermore, we have shown that these compounds inhibit PPI of HER2:HER3 and phosphorylation of HER2 as compared to control in tissue samples derived from in vivo studies. The stability of the compounds was also investigated in mouse serum, and the compounds exhibited stability with a half-life of up to 3 h. These results suggest that the novel peptidomimetics we have developed target the extracellular domain of HER2 protein and inhibit HER2:HER3 interaction, providing a novel method to treat HER2-positive cancer.
Keywords: HER2; HER3; breast cancer; peptidomimetic; protein-protein interaction.
© 2014 John Wiley & Sons A/S.
Figures


Similar articles
-
Design of novel lipidated peptidomimetic conjugates for targeting EGFR heterodimerization in HER2 + cancer.Bioorg Med Chem Lett. 2018 Dec 1;28(22):3506-3513. doi: 10.1016/j.bmcl.2018.10.005. Epub 2018 Oct 3. Bioorg Med Chem Lett. 2018. PMID: 30314880 Free PMC article.
-
Structure-activity relationships of peptidomimetics that inhibit PPI of HER2-HER3.Biopolymers. 2014 Jun;101(6):693-702. doi: 10.1002/bip.22441. Biopolymers. 2014. PMID: 24222531 Free PMC article.
-
Design of cyclic and d-amino acids containing peptidomimetics for inhibition of protein-protein interactions of HER2-HER3.J Pept Sci. 2018 Feb;24(2):10.1002/psc.3066. doi: 10.1002/psc.3066. J Pept Sci. 2018. PMID: 29436155 Free PMC article.
-
Mechanisms of acquired resistance to HER2-Positive breast cancer therapies induced by HER3: A comprehensive review.Eur J Pharmacol. 2024 Aug 15;977:176725. doi: 10.1016/j.ejphar.2024.176725. Epub 2024 Jun 6. Eur J Pharmacol. 2024. PMID: 38851563 Review.
-
Pertuzumab: optimizing HER2 blockade.Clin Cancer Res. 2013 Oct 15;19(20):5552-6. doi: 10.1158/1078-0432.CCR-13-0518. Epub 2013 Aug 13. Clin Cancer Res. 2013. PMID: 23942091 Review.
Cited by
-
Signaling pathways and therapeutic interventions in gastric cancer.Signal Transduct Target Ther. 2022 Oct 8;7(1):358. doi: 10.1038/s41392-022-01190-w. Signal Transduct Target Ther. 2022. PMID: 36209270 Free PMC article. Review.
-
In vivo studies of a peptidomimetic that targets EGFR dimerization in NSCLC.J Cancer. 2020 Aug 18;11(20):5982-5999. doi: 10.7150/jca.46320. eCollection 2020. J Cancer. 2020. PMID: 32922539 Free PMC article.
-
A grafted peptidomimetic for EGFR heterodimerization inhibition: Implications in NSCLC models.Eur J Med Chem. 2021 Apr 15;216:113312. doi: 10.1016/j.ejmech.2021.113312. Epub 2021 Feb 23. Eur J Med Chem. 2021. PMID: 33667849 Free PMC article.
-
Design of novel lipidated peptidomimetic conjugates for targeting EGFR heterodimerization in HER2 + cancer.Bioorg Med Chem Lett. 2018 Dec 1;28(22):3506-3513. doi: 10.1016/j.bmcl.2018.10.005. Epub 2018 Oct 3. Bioorg Med Chem Lett. 2018. PMID: 30314880 Free PMC article.
-
Emerging anti-cancer antibodies and combination therapies targeting HER3/ERBB3.Hum Vaccin Immunother. 2016 Mar 3;12(3):576-92. doi: 10.1080/21645515.2015.1102809. Epub 2015 Nov 3. Hum Vaccin Immunother. 2016. PMID: 26529100 Free PMC article. Review.
References
-
- Molina MA, Codony-Servat J, Albanell J, Rojo F, Arribas J, Baselga J. Trastuzumab (herceptin), a humanized anti-Her2 receptor monoclonal antibody, inhibits basal and activated Her2 ectodomain cleavage in breast cancer cells. Cancer Res. 2001;61:4744–9. - PubMed
-
- Slamon DJ. Proto-oncogenes and human cancers. N Engl J Med. 1987;317:955–7. - PubMed
-
- Tebbutt N, Pedersen MW, Johns TG. Targeting the ERBB family in cancer: couples therapy. Nat Rev Cancer. 2013;13:663–73. - PubMed
-
- Chang JC. HER2 inhibition: from discovery to clinical practice. Clinical Cancer Res. 2007;13:1–3. - PubMed
-
- Niehans GA, Singleton TP, Dykoski D, Kiang DT. Stability of HER-2/neu expression over time and at multiple metastatic sites. J Natl Cancer Inst. 1993;85:1230–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

