Longitudinal changes in resting-state brain activity in a capsular infarct model
- PMID: 25352047
- PMCID: PMC4814054
- DOI: 10.1038/jcbfm.2014.178
Longitudinal changes in resting-state brain activity in a capsular infarct model
Erratum in
-
Longitudinal changes in resting-state brain activity in a capsular infarct model.J Cereb Blood Flow Metab. 2015 May;35(5):882. doi: 10.1038/jcbfm.2015.15. J Cereb Blood Flow Metab. 2015. PMID: 25924830 Free PMC article. No abstract available.
Abstract
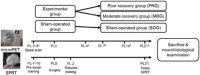
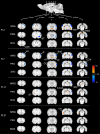
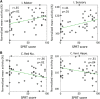
Strokes attributable to subcortical infarcts have been increasing recently in elderly patients. To gain insight how this lesion influences the motor outcome and responds to rehabilitative training, we used circumscribed photothrombotic capsular infarct models on 36 Sprague-Dawley rats (24 experimental and 12 sham-operated). We used 2-deoxy-2-[(18)F]-fluoro-D-glucose-micro positron emission tomography (FDG-microPET) to assess longitudinal changes in resting-state brain activity (rs-BA) and daily single-pellet reaching task (SPRT) trainings to evaluate motor recovery. Longitudinal FDG-microPET results showed that capsular infarct resulted in a persistent decrease in rs-BA in bilateral sensory and auditory cortices, and ipsilesional motor cortex, thalamus, and inferior colliculus (P<0.0025, false discovery rate (FDR) q<0.05). The decreased rs-BA is compatible with diaschisis and contributes to manifest the malfunctions of lesion-specific functional connectivity. In contrast, capsular infarct resulted in increase of rs-BA in the ipsilesional internal capsule, and contralesional red nucleus and ventral hippocampus in recovery group (P<0.0025, FDR q<0.05), implying that remaining subcortical structures have an important role in conducting the recovery process in capsular infarct. The SPRT training facilitated motor recovery only in rats with an incomplete destruction of the posterior limb of the internal capsule (PLIC) (Pearson's correlation, P<0.05). Alternative therapeutic interventions are required to enhance the potential for recovery in capsular infarct with complete destruction of PLIC.
Figures



References
-
- Bamford J, Sandercock P, Dennis M, Burn J, Warlow C. Classification and natural history of clinically identifiable subtypes of cerebral infarction. Lancet 1991; 337: 1521–1526. - PubMed
-
- Lecrux C, McCabe C, Weir CJ, Gallagher L, Mullin J, Touzani O et al. Effects of magnesium treatment in a model of internal capsule lesion in spontaneously hypertensive rats. Stroke 2008; 39: 448–454. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

