Cardiomyocyte-specific deficiency of ketone body metabolism promotes accelerated pathological remodeling
- PMID: 25353003
- PMCID: PMC4209361
- DOI: 10.1016/j.molmet.2014.07.010
Cardiomyocyte-specific deficiency of ketone body metabolism promotes accelerated pathological remodeling
Abstract
Objective: Exploitation of protective metabolic pathways within injured myocardium still remains an unclarified therapeutic target in heart disease. Moreover, while the roles of altered fatty acid and glucose metabolism in the failing heart have been explored, the influence of highly dynamic and nutritionally modifiable ketone body metabolism in the regulation of myocardial substrate utilization, mitochondrial bioenergetics, reactive oxygen species (ROS) generation, and hemodynamic response to injury remains undefined.
Methods: Here we use mice that lack the enzyme required for terminal oxidation of ketone bodies, succinyl-CoA:3-oxoacid CoA transferase (SCOT) to determine the role of ketone body oxidation in the myocardial injury response. Tracer delivery in ex vivo perfused hearts coupled to NMR spectroscopy, in vivo high-resolution echocardiographic quantification of cardiac hemodynamics in nutritionally and surgically modified mice, and cellular and molecular measurements of energetic and oxidative stress responses are performed.
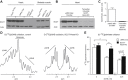
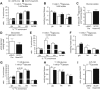
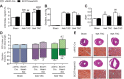
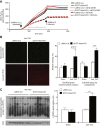
Results: While germline SCOT-knockout (KO) mice die in the early postnatal period, adult mice with cardiomyocyte-specific loss of SCOT (SCOT-Heart-KO) remarkably exhibit no overt metabolic abnormalities, and no differences in left ventricular mass or impairments of systolic function during periods of ketosis, including fasting and adherence to a ketogenic diet. Myocardial fatty acid oxidation is increased when ketones are delivered but cannot be oxidized. To determine the role of ketone body oxidation in the remodeling ventricle, we induced pressure overload injury by performing transverse aortic constriction (TAC) surgery in SCOT-Heart-KO and αMHC-Cre control mice. While TAC increased left ventricular mass equally in both groups, at four weeks post-TAC, myocardial ROS abundance was increased in myocardium of SCOT-Heart-KO mice, and mitochondria and myofilaments were ultrastructurally disordered. Eight weeks post-TAC, left ventricular volume was markedly increased and ejection fraction was decreased in SCOT-Heart-KO mice, while these parameters remained normal in hearts of control animals.
Conclusions: These studies demonstrate the ability of myocardial ketone metabolism to coordinate the myocardial response to pressure overload, and suggest that the oxidation of ketone bodies may be an important contributor to free radical homeostasis and hemodynamic preservation in the injured heart.
Keywords: Mitochondrial metabolism; Myocardial ketone body metabolism; Nuclear magnetic resonance (NMR) measurement of substrate metabolism; Oxidative stress; Ventricular remodeling.
Figures



References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

