Airway basal cells. The "smoking gun" of chronic obstructive pulmonary disease
- PMID: 25354273
- PMCID: PMC4299651
- DOI: 10.1164/rccm.201408-1492PP
Airway basal cells. The "smoking gun" of chronic obstructive pulmonary disease
Abstract
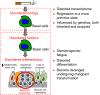
The earliest abnormality in the lung associated with smoking is hyperplasia of airway basal cells, the stem/progenitor cells of the ciliated and secretory cells that are central to pulmonary host defense. Using cell biology and 'omics technologies to assess basal cells isolated from bronchoscopic brushings of nonsmokers, smokers, and smokers with chronic obstructive pulmonary disease (COPD), compelling evidence has been provided in support of the concept that airway basal cells are central to the pathogenesis of smoking-associated lung diseases. When confronted by the chronic stress of smoking, airway basal cells become disorderly, regress to a more primitive state, behave as dictated by their inheritance, are susceptible to acquired changes in their genome, lose the capacity to regenerate the epithelium, are responsible for the major changes in the airway that characterize COPD, and, with persistent stress, can undergo malignant transformation. Together, these observations led to the conclusion that accelerated loss of lung function in susceptible individuals begins with disordered airway basal cell biology (i.e., that airway basal cells are the "smoking gun" of COPD, a potential target for the development of therapies to prevent smoking-related lung disorders).
Keywords: COPD; airway epithelium; basal cells; lung cancer; smoking.
Figures


References
-
- Faner R, Tal-Singer R, Riley JH, Celli B, Vestbo J, MacNee W, Bakke P, Calverley PM, Coxson H, Crim C, et al. ECLIPSE Study Investigators. Lessons from ECLIPSE: a review of COPD biomarkers. Thorax. 2014;69:666–672. - PubMed
-
- Yoshida T, Tuder RM. Pathobiology of cigarette smoke-induced chronic obstructive pulmonary disease. Physiol Rev. 2007;87:1047–1082. - PubMed
-
- Auerbach O, Stout AP, Hammond EC, Garfinkel L. Changes in bronchial epithelium in relation to cigarette smoking and in relation to lung cancer. N Engl J Med. 1961;265:253–267. - PubMed
-
- Hogg JC, Chu F, Utokaparch S, Woods R, Elliott WM, Buzatu L, Cherniack RM, Rogers RM, Sciurba FC, Coxson HO, et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N Engl J Med. 2004;350:2645–2653. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

