Pancreatic Cancer-Derived Exosomes Cause Paraneoplastic β-cell Dysfunction
- PMID: 25355928
- PMCID: PMC4383684
- DOI: 10.1158/1078-0432.CCR-14-2022
Pancreatic Cancer-Derived Exosomes Cause Paraneoplastic β-cell Dysfunction
Erratum in
-
Correction: Pancreatic Cancer-Derived Exosomes Causes Paraneoplastic β-cell Dysfunction.Clin Cancer Res. 2015 Oct 1;21(19):4495. doi: 10.1158/1078-0432.CCR-15-1524. Clin Cancer Res. 2015. PMID: 26429984 No abstract available.
Abstract
Purpose: Pancreatic cancer frequently causes diabetes. We recently proposed adrenomedullin as a candidate mediator of pancreatic β-cell dysfunction in pancreatic cancer. How pancreatic cancer-derived adrenomedullin reaches β cells remote from the cancer to induce β-cell dysfunction is unknown. We tested a novel hypothesis that pancreatic cancer sheds adrenomedullin-containing exosomes into circulation, which are transported to β cells and impair insulin secretion.
Experimental methods: We characterized exosomes from conditioned media of pancreatic cancer cell lines (n = 5) and portal/peripheral venous blood of patients with pancreatic cancer (n = 20). Western blot analysis showed the presence of adrenomedullin in pancreatic cancer-exosomes. We determined the effect of adrenomedullin-containing pancreatic cancer exosomes on insulin secretion from INS-1 β cells and human islets, and demonstrated the mechanism of exosome internalization into β cells. We studied the interaction between β-cell adrenomedullin receptors and adrenomedullin present in pancreatic cancer-exosomes. In addition, the effect of adrenomedullin on endoplasmic reticulum (ER) stress response genes and reactive oxygen/nitrogen species generation in β cells was shown.
Results: Exosomes were found to be the predominant extracellular vesicles secreted by pancreatic cancer into culture media and patient plasma. Pancreatic cancer-exosomes contained adrenomedullin and CA19-9, readily entered β cells through caveolin-mediated endocytosis or macropinocytosis, and inhibited insulin secretion. Adrenomedullin in pancreatic cancer exosomes interacted with its receptor on β cells. Adrenomedullin receptor blockade abrogated the inhibitory effect of exosomes on insulin secretion. β cells exposed to adrenomedullin or pancreatic cancer exosomes showed upregulation of ER stress genes and increased reactive oxygen/nitrogen species.
Conclusions: Pancreatic cancer causes paraneoplastic β-cell dysfunction by shedding adrenomedullin(+)/CA19-9(+) exosomes into circulation that inhibit insulin secretion, likely through adrenomedullin-induced ER stress and failure of the unfolded protein response.
©2014 American Association for Cancer Research.
Conflict of interest statement
Figures


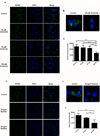


Comment in
-
Pancreatic cancer-associated diabetes is an "exosomopathy".Clin Cancer Res. 2015 Apr 1;21(7):1508-10. doi: 10.1158/1078-0432.CCR-14-2990. Epub 2015 Feb 2. Clin Cancer Res. 2015. PMID: 25645860 Free PMC article.
References
-
- Matrisian LMRA. PANCAN. The alarming rise of pancreatic cancer deaths in the United States: Why we need to stem the tide. Pancreatic Cancer Action network. 2013
-
- Permert J, Ihse I, Jorfeldt L, von Schenck H, Arnqvist HJ, Larsson J. Pancreatic cancer is associated with impaired glucose metabolism. Eur J Surg. 1993;159:101–107. - PubMed
-
- Chari ST, Klee GG, Miller LJ, Raimondo M, DiMagno EP. Islet amyloid polypeptide is not a satisfactory marker for detecting pancreatic cancer. Gastroenterology. 2001;121:640–645. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 DK088227/DK/NIDDK NIH HHS/United States
- DK088227/DK/NIDDK NIH HHS/United States
- DK042394/DK/NIDDK NIH HHS/United States
- 1T32 CA148073/CA/NCI NIH HHS/United States
- HL052173/HL/NHLBI NIH HHS/United States
- R29 CA078383/CA/NCI NIH HHS/United States
- P30 CA030199/CA/NCI NIH HHS/United States
- P50 CA102701/CA/NCI NIH HHS/United States
- CA78383/CA/NCI NIH HHS/United States
- P30 CA015083/CA/NCI NIH HHS/United States
- R01 CA078383/CA/NCI NIH HHS/United States
- R01 HL052173/HL/NHLBI NIH HHS/United States
- T32 CA148073/CA/NCI NIH HHS/United States
- CA150190/CA/NCI NIH HHS/United States
- R24 DK093074/DK/NIDDK NIH HHS/United States
- CA100685/CA/NCI NIH HHS/United States
- R01 CA150190/CA/NCI NIH HHS/United States
- R01 CA100685/CA/NCI NIH HHS/United States
- P30 DK084567/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

