AMG 900, a potent inhibitor of aurora kinases causes pharmacodynamic changes in p-Histone H3 immunoreactivity in human tumor xenografts and proliferating mouse tissues
- PMID: 25367255
- PMCID: PMC4221688
- DOI: 10.1186/s12967-014-0307-x
AMG 900, a potent inhibitor of aurora kinases causes pharmacodynamic changes in p-Histone H3 immunoreactivity in human tumor xenografts and proliferating mouse tissues
Abstract
Background: The Aurora family of serine-threonine kinases are essential regulators of cell division in mammalian cells. Aurora-A and -B expression and kinase activity is elevated in a variety of human cancers and is associated with high proliferation rates and poor prognosis. AMG 900 is a highly potent and selective pan-aurora kinase inhibitor that has entered clinical evaluation in adult patients with advanced cancers. In mice, oral administration of AMG 900 blocks the phosphorylation of histone H3 on serine-10 (p-Histone H3), a proximal substrate of aurora-B and inhibits the growth of multiple human tumor xenografts, including multidrug-resistant models.
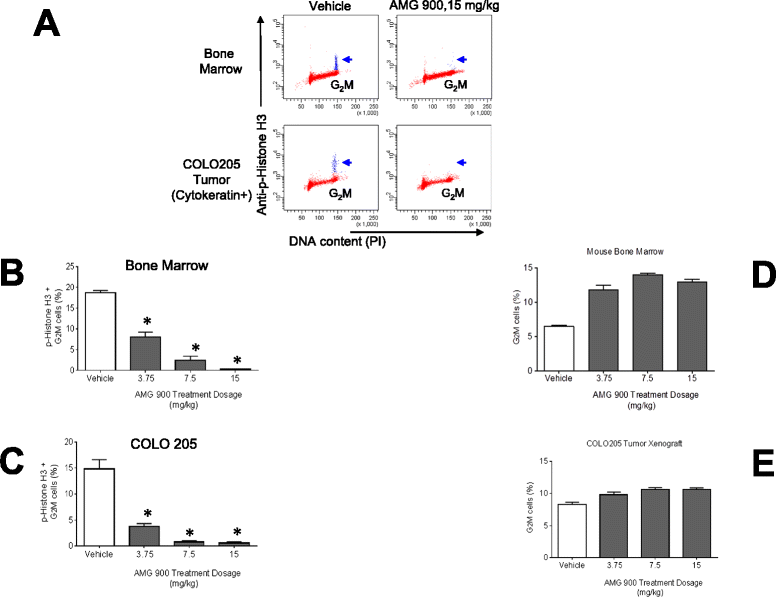
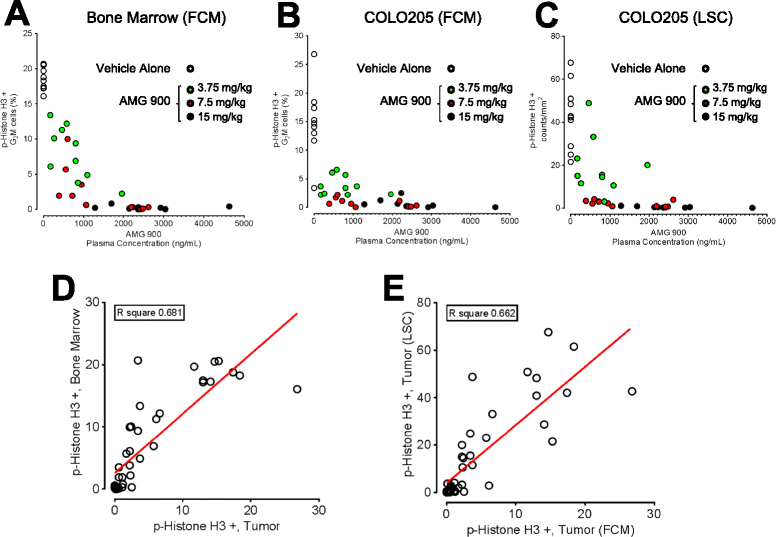
Methods: In order to establish a preclinical pharmacokinetic-pharmacodynamic (PK-PD) relationship for AMG 900 that could be translated to the clinic, we used flow cytometry and laser scanning cytometry detection platforms to assess the effects on p-Histone H3 inhibition in terms of sensitivity, precision, and specificity, in human tumor xenografts in conjunction with mouse skin and bone marrow tissues. Mice with established COLO 205 tumors were administered AMG 900 at 3.75, 7.5, and 15 mg/kg and assessed after 3 hours.
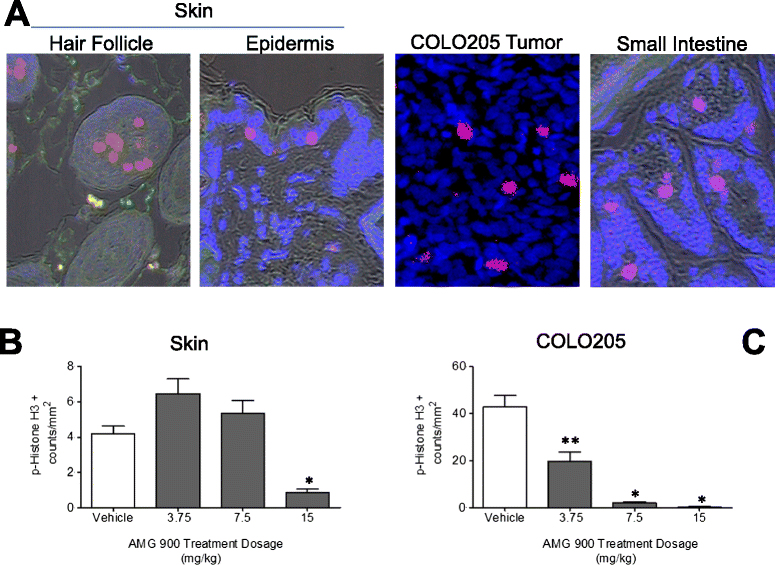
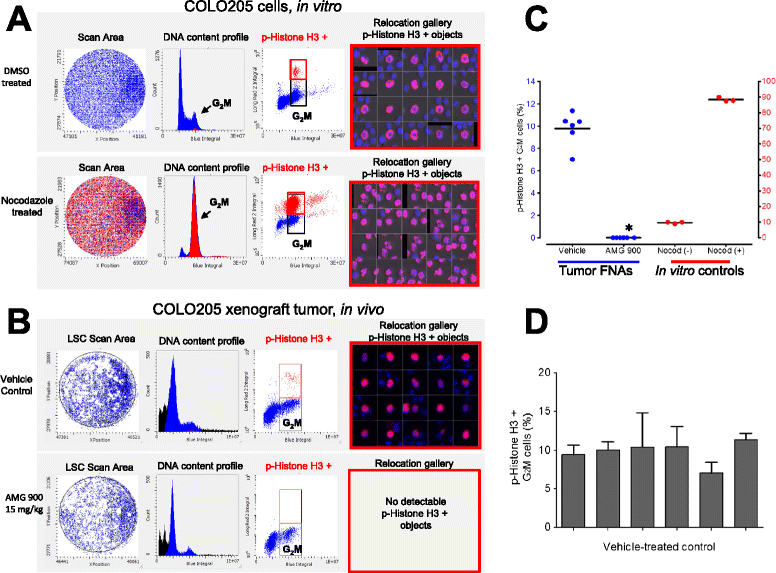
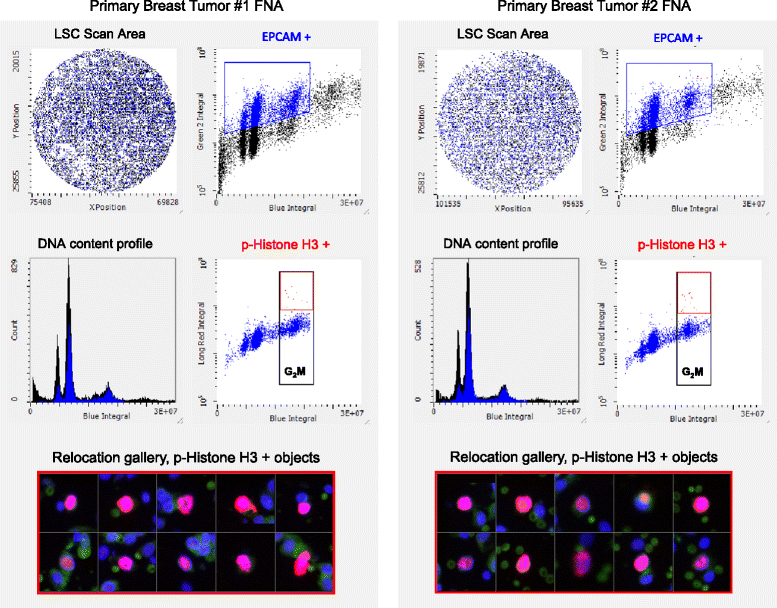
Results: Significant suppression of p-Histone H3 in mouse skin was only observed at 15 mg/kg (p <0.0001), whereas in mouse bone marrow and in tumor a dose-dependent inhibition was achieved at all three doses (p ≤ 0.00015). These studies demonstrate that AMG 900 inhibits p-Histone H3 in tumors and surrogate tissues (although tissues such as skin may be less sensitive for assessing PD effects). To further extend our work, we evaluated the feasibility of measuring p-Histone H3 using fine-needle aspirate (FNA) tumor xenograft biopsies. Treatment with AMG 900 significantly inhibited p-Histone H3 (>99% inhibition, p <0.0001) in COLO 205 tumors. Lastly, we illustrate this LSC-based approach can detect p-Histone H3 positive cells using mock FNAs from primary human breast tumor tissues.
Conclusion: Phosphorylation of histone H3 is a useful biomarker to determine the pharmacodynamics (PD) activity of AMG 900. FNA biopsies may be a viable approach for assessing AMG 900 PD effects in the clinic.
Figures
References
-
- Gizatullin F, Yao Y, Kung V, Harding MW, Loda M, Shapiro GI. The aurora kinase inhibitor VX-680 induces endoreduplication and apoptosis preferentially in cells with compromised p53-dependent postmitiotic checkpoint function. Cancer Res. 2006;66:7668–7677. doi: 10.1158/0008-5472.CAN-05-3353. - DOI - PubMed
-
- Harrington EA, Bebbington D, Moore J, Rasmussen RK, Ajose-Adeogun AO, Nakayama T, Graham JA, Demur C, Hercend T, Diu-Hercend A, Su M, Golec JMC, Miller KM. VX-680, a potent and selective small-molecule inhibitor of the aurora kinases, suppresses tumor growth in vivo. Nat Med. 2004;10:262–267. doi: 10.1038/nm1003. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

