Host intestinal signal-promoted biofilm dispersal induces Vibrio cholerae colonization
- PMID: 25368110
- PMCID: PMC4288906
- DOI: 10.1128/IAI.02617-14
Host intestinal signal-promoted biofilm dispersal induces Vibrio cholerae colonization
Abstract
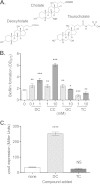
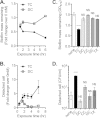
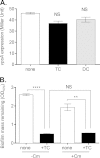
Vibrio cholerae causes human infection through ingestion of contaminated food and water, leading to the devastating diarrheal disease cholera. V. cholerae forms matrix-encased aggregates, known as biofilms, in the native aquatic environment. While the formation of V. cholerae biofilms has been well studied, little is known about the dispersal from biofilms, particularly upon entry into the host. In this study, we found that the exposure of mature biofilms to physiologic levels of the bile salt taurocholate, a host signal for the virulence gene induction of V. cholerae, induces an increase in the number of detached cells with a concomitant decrease in biofilm mass. Scanning electron microscopy micrographs of biofilms exposed to taurocholate revealed an altered, perhaps degraded, appearance of the biofilm matrix. The inhibition of protein synthesis did not alter rates of detachment, suggesting that V. cholerae undergoes a passive dispersal. Cell-free media from taurocholate-exposed biofilms contains a larger amount of free polysaccharide, suggesting an abiotic degradation of biofilm matrix by taurocholate. Furthermore, we found that V. cholerae is only able to induce virulence in response to taurocholate after exit from the biofilm. Thus, we propose a model in which V. cholerae ingested as a biofilm has coopted the host-derived bile salt signal to detach from the biofilm and go on to activate virulence.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures



Similar articles
-
Living in the matrix: assembly and control of Vibrio cholerae biofilms.Nat Rev Microbiol. 2015 May;13(5):255-68. doi: 10.1038/nrmicro3433. Nat Rev Microbiol. 2015. PMID: 25895940 Free PMC article. Review.
-
Vibrio cholerae Biofilms and Cholera Pathogenesis.PLoS Negl Trop Dis. 2016 Feb 4;10(2):e0004330. doi: 10.1371/journal.pntd.0004330. eCollection 2016 Feb. PLoS Negl Trop Dis. 2016. PMID: 26845681 Free PMC article. Review.
-
Identification of signaling pathways, matrix-digestion enzymes, and motility components controlling Vibrio cholerae biofilm dispersal.Proc Natl Acad Sci U S A. 2020 Dec 22;117(51):32639-32647. doi: 10.1073/pnas.2021166117. Epub 2020 Dec 7. Proc Natl Acad Sci U S A. 2020. PMID: 33288715 Free PMC article.
-
The virulence transcriptional activator AphA enhances biofilm formation by Vibrio cholerae by activating expression of the biofilm regulator VpsT.Infect Immun. 2010 Feb;78(2):697-703. doi: 10.1128/IAI.00429-09. Epub 2009 Nov 23. Infect Immun. 2010. PMID: 19933826 Free PMC article.
-
Quorum sensing-dependent biofilms enhance colonization in Vibrio cholerae.Dev Cell. 2003 Oct;5(4):647-56. doi: 10.1016/s1534-5807(03)00295-8. Dev Cell. 2003. PMID: 14536065
Cited by
-
Living in the matrix: assembly and control of Vibrio cholerae biofilms.Nat Rev Microbiol. 2015 May;13(5):255-68. doi: 10.1038/nrmicro3433. Nat Rev Microbiol. 2015. PMID: 25895940 Free PMC article. Review.
-
Intestinal Colonization Dynamics of Vibrio cholerae.PLoS Pathog. 2015 May 21;11(5):e1004787. doi: 10.1371/journal.ppat.1004787. eCollection 2015 May. PLoS Pathog. 2015. PMID: 25996593 Free PMC article. Review.
-
Mechanisms of Bacterial Tolerance and Persistence in the Gastrointestinal and Respiratory Environments.Clin Microbiol Rev. 2018 Aug 1;31(4):e00023-18. doi: 10.1128/CMR.00023-18. Print 2018 Oct. Clin Microbiol Rev. 2018. PMID: 30068737 Free PMC article. Review.
-
Diguanylate Cyclases in Vibrio cholerae: Essential Regulators of Lifestyle Switching.Front Cell Infect Microbiol. 2020 Oct 22;10:582947. doi: 10.3389/fcimb.2020.582947. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 33194821 Free PMC article. Review.
-
A mannose-sensing AraC-type transcriptional activator regulates cell-cell aggregation of Vibrio cholerae.NPJ Biofilms Microbiomes. 2022 Aug 20;8(1):65. doi: 10.1038/s41522-022-00331-x. NPJ Biofilms Microbiomes. 2022. PMID: 35987769 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

