Coronavirus cell entry occurs through the endo-/lysosomal pathway in a proteolysis-dependent manner
- PMID: 25375324
- PMCID: PMC4223067
- DOI: 10.1371/journal.ppat.1004502
Coronavirus cell entry occurs through the endo-/lysosomal pathway in a proteolysis-dependent manner
Erratum in
-
Correction: Coronavirus cell entry occurs through the endo-/lysosomal pathway in a proteolysis-dependent manner.PLoS Pathog. 2015 Feb 13;11(2):e1004709. doi: 10.1371/journal.ppat.1004709. eCollection 2015 Feb. PLoS Pathog. 2015. PMID: 25679792 Free PMC article.
Abstract
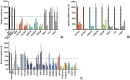
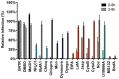
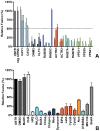
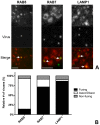
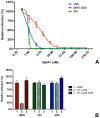
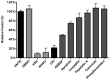
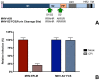
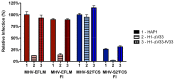
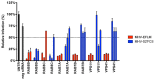
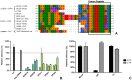
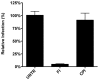
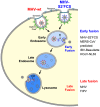
Enveloped viruses need to fuse with a host cell membrane in order to deliver their genome into the host cell. While some viruses fuse with the plasma membrane, many viruses are endocytosed prior to fusion. Specific cues in the endosomal microenvironment induce conformational changes in the viral fusion proteins leading to viral and host membrane fusion. In the present study we investigated the entry of coronaviruses (CoVs). Using siRNA gene silencing, we found that proteins known to be important for late endosomal maturation and endosome-lysosome fusion profoundly promote infection of cells with mouse hepatitis coronavirus (MHV). Using recombinant MHVs expressing reporter genes as well as a novel, replication-independent fusion assay we confirmed the importance of clathrin-mediated endocytosis and demonstrated that trafficking of MHV to lysosomes is required for fusion and productive entry to occur. Nevertheless, MHV was shown to be less sensitive to perturbation of endosomal pH than vesicular stomatitis virus and influenza A virus, which fuse in early and late endosomes, respectively. Our results indicate that entry of MHV depends on proteolytic processing of its fusion protein S by lysosomal proteases. Fusion of MHV was severely inhibited by a pan-lysosomal protease inhibitor, while trafficking of MHV to lysosomes and processing by lysosomal proteases was no longer required when a furin cleavage site was introduced in the S protein immediately upstream of the fusion peptide. Also entry of feline CoV was shown to depend on trafficking to lysosomes and processing by lysosomal proteases. In contrast, MERS-CoV, which contains a minimal furin cleavage site just upstream of the fusion peptide, was negatively affected by inhibition of furin, but not of lysosomal proteases. We conclude that a proteolytic cleavage site in the CoV S protein directly upstream of the fusion peptide is an essential determinant of the intracellular site of fusion.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
References
-
- Okada Y (1969) Factors in fusion of cells by HVJ. Current topics in microbiology and immunology 48: 102–128. - PubMed
-
- Permanyer M, Ballana E, Este JA (2010) Endocytosis of HIV: anything goes. Trends in microbiology 18: 543–551. - PubMed
-
- Stein BS, Gowda SD, Lifson JD, Penhallow RC, Bensch KG, et al. (1987) pH-independent HIV entry into CD4-positive T cells via virus envelope fusion to the plasma membrane. Cell 49: 659–668. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

