Cerebellar-dependent expression of motor learning during eyeblink conditioning in head-fixed mice
- PMID: 25378152
- PMCID: PMC4220020
- DOI: 10.1523/JNEUROSCI.2820-14.2014
Cerebellar-dependent expression of motor learning during eyeblink conditioning in head-fixed mice
Abstract
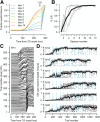
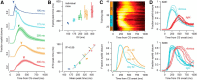
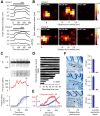
Eyeblink conditioning in restrained rabbits has served as an excellent model of cerebellar-dependent motor learning for many decades. In mice, the role of the cerebellum in eyeblink conditioning is less clear and remains controversial, partly because learning appears to engage fear-related circuits and lesions of the cerebellum do not abolish the learned behavior completely. Furthermore, experiments in mice are performed using freely moving systems, which lack the stability necessary for mapping out the essential neural circuitry with electrophysiological approaches. We have developed a novel apparatus for eyeblink conditioning in head-fixed mice. Here, we show that the performance of mice in our apparatus is excellent and that the learned behavior displays two hallmark features of cerebellar-dependent eyeblink conditioning in rabbits: (1) gradual acquisition; and (2) adaptive timing of conditioned movements. Furthermore, we use a combination of pharmacological inactivation, electrical stimulation, single-unit recordings, and targeted microlesions to demonstrate that the learned behavior is completely dependent on the cerebellum and to pinpoint the exact location in the deep cerebellar nuclei that is necessary. Our results pave the way for using eyeblink conditioning in head-fixed mice as a platform for applying next-generation genetic tools to address molecular and circuit-level questions about cerebellar function in health and disease.
Keywords: cerebellum; classical conditioning; interpositus; mouse; neurophysiology; timing.
Copyright © 2014 the authors 0270-6474/14/3414845-09$15.00/0.
Figures



Similar articles
-
Impaired classical eyeblink conditioning in cerebellar-lesioned and Purkinje cell degeneration (pcd) mutant mice.J Neurosci. 1996 Apr 15;16(8):2829-38. doi: 10.1523/JNEUROSCI.16-08-02829.1996. J Neurosci. 1996. PMID: 8786457 Free PMC article.
-
Changes in rabbit cerebellar cortical and interpositus nucleus activity during acquisition, extinction, and backward classical eyelid conditioning.Neurobiol Learn Mem. 1996 Jan;65(1):17-34. doi: 10.1006/nlme.1996.0003. Neurobiol Learn Mem. 1996. PMID: 8673404
-
In vitro classical conditioning of the turtle eyeblink reflex: approaching cellular mechanisms of acquisition.Cerebellum. 2003;2(1):55-61. doi: 10.1080/14734220310015610. Cerebellum. 2003. PMID: 12882235
-
Ontogenetic changes in the neural mechanisms of eyeblink conditioning.Integr Physiol Behav Sci. 2001 Jan-Mar;36(1):15-35. doi: 10.1007/BF02733945. Integr Physiol Behav Sci. 2001. PMID: 11484994 Review.
-
Cerebellar circuits and synaptic mechanisms involved in classical eyeblink conditioning.Trends Neurosci. 1997 Apr;20(4):177-81. doi: 10.1016/s0166-2236(96)10081-3. Trends Neurosci. 1997. PMID: 9106359 Review.
Cited by
-
Whisker-signaled Eyeblink Classical Conditioning in Head-fixed Mice.J Vis Exp. 2016 Mar 30;(109):e53310. doi: 10.3791/53310. J Vis Exp. 2016. PMID: 27077752 Free PMC article.
-
Synaptic variance and action potential firing of cerebellar output neurons during motor learning in larval zebrafish.Curr Biol. 2023 Aug 21;33(16):3299-3311.e3. doi: 10.1016/j.cub.2023.06.045. Epub 2023 Jul 7. Curr Biol. 2023. PMID: 37421952 Free PMC article.
-
Visual Sequences Drive Experience-Dependent Plasticity in Mouse Anterior Cingulate Cortex.Cell Rep. 2020 Sep 15;32(11):108152. doi: 10.1016/j.celrep.2020.108152. Cell Rep. 2020. PMID: 32937128 Free PMC article.
-
Chromatin remodeling inactivates activity genes and regulates neural coding.Science. 2016 Jul 15;353(6296):300-305. doi: 10.1126/science.aad4225. Science. 2016. PMID: 27418512 Free PMC article.
-
A Flexible Platform for Monitoring Cerebellum-Dependent Sensory Associative Learning.J Vis Exp. 2022 Jan 19;(179):10.3791/63205. doi: 10.3791/63205. J Vis Exp. 2022. PMID: 35129170 Free PMC article.
References
-
- Berthier NE, Moore JW. Activity of deep cerebellar nuclear cells during classical conditioning of nictitating membrane extension in rabbits. Exp Brain Res. 1990;83:44–54. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
