Serine-arginine protein kinase 1 (SRPK1) inhibition as a potential novel targeted therapeutic strategy in prostate cancer
- PMID: 25381816
- PMCID: PMC4351909
- DOI: 10.1038/onc.2014.360
Serine-arginine protein kinase 1 (SRPK1) inhibition as a potential novel targeted therapeutic strategy in prostate cancer
Abstract
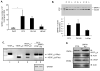
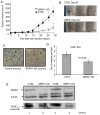
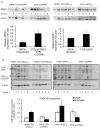
Angiogenesis is required for tumour growth and is induced principally by vascular endothelial growth factor A (VEGF-A). VEGF-A pre-mRNA is alternatively spliced at the terminal exon to produce two families of isoforms, pro- and anti-angiogenic, only the former of which is upregulated in prostate cancer (PCa). In renal epithelial cells and colon cancer cells, the choice of VEGF splice isoforms is controlled by the splicing factor SRSF1, phosphorylated by serine-arginine protein kinase 1 (SRPK1). Immunohistochemistry staining of human samples revealed a significant increase in SRPK1 expression both in prostate intra-epithelial neoplasia lesions as well as malignant adenocarcinoma compared with benign prostate tissue. We therefore tested the hypothesis that the selective upregulation of pro-angiogenic VEGF in PCa may be under the control of SRPK1 activity. A switch in the expression of VEGF165 towards the anti-angiogenic splice isoform, VEGF165b, was seen in PC-3 cells with SRPK1 knockdown (KD). PC-3 SRPK1-KD cells resulted in tumours that grew more slowly in xenografts, with decreased microvessel density. No effect was seen as a result of SRPK1-KD on growth, proliferation, migration and invasion capabilities of PC-3 cells in vitro. Small-molecule inhibitors of SRPK1 switched splicing towards the anti-angiogenic isoform VEGF165b in PC-3 cells and decreased tumour growth when administered intraperitoneally in an orthotopic mouse model of PCa. Our study suggests that modulation of SRPK1 and subsequent inhibition of tumour angiogenesis by regulation of VEGF splicing can alter prostate tumour growth and supports further studies for the use of SRPK1 inhibition as a potential anti-angiogenic therapy in PCa.
Figures




References
-
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013 Jan;63(1):11–30. - PubMed
-
- Gatta G, Mallone S, van der Zwan JM, Trama A, Siesling S, Capocaccia R. Cancer prevalence estimates in Europe at the beginning of 2000. Ann Oncol. 2013 Jun;24(6):1660–6. - PubMed
-
- Quaglia A, Lillini R, Crocetti E, Buzzoni C, Vercelli M. Incidence and mortality trends for four major cancers in the elderly and middle-aged adults: An international comparison. Surg Oncol. 2013 Mar 24; - PubMed
-
- Malvezzi M, Bertuccio P, Levi F, La Vecchia C, Negri E. European cancer mortality predictions for the year 2013. Ann Oncol. 2013 Mar;24(3):792–800. - PubMed
-
- Whang YE, Armstrong AJ, Rathmell WK, Godley PA, Kim WY, Pruthi RS, et al. A phase II study of lapatinib, a dual EGFR and HER-2 tyrosine kinase inhibitor, in patients with castration-resistant prostate cancer. Urol Oncol. 2013 Jan;31(1):82–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- G10002073/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- C11392/A10484/CRUK_/Cancer Research UK/United Kingdom
- G1002073/MRC_/Medical Research Council/United Kingdom
- MR/K020366/1/MRC_/Medical Research Council/United Kingdom
- BB/J007293/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

