Shear stress-induced NO production is dependent on ATP autocrine signaling and capacitative calcium entry
- PMID: 25386222
- PMCID: PMC4224574
- DOI: 10.1007/s12195-014-0351-x
Shear stress-induced NO production is dependent on ATP autocrine signaling and capacitative calcium entry
Abstract
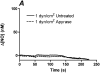
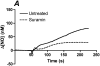
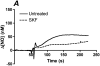
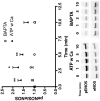
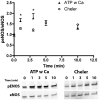
Flow-induced production of nitric oxide (NO) by endothelial cells plays a fundamental role in vascular homeostasis. However, the mechanisms by which shear stress activates NO production remain unclear due in part to limitations in measuring NO, especially under flow conditions. Shear stress elicits the release of ATP, but the relative contribution of autocrine stimulation by ATP to flow-induced NO production has not been established. Furthermore, the importance of calcium in shear stress-induced NO production remains controversial, and in particular the role of capacitive calcium entry (CCE) has yet to be determined. We have utilized our unique NO measurement device to investigate the role of ATP autocrine signaling and CCE in shear stress-induced NO production. We found that endogenously released ATP and downstream activation of purinergic receptors and CCE plays a significant role in shear stress-induced NO production. ATP-induced eNOS phophorylation under static conditions is also dependent on CCE. Inhibition of protein kinase C significantly inhibited eNOS phosphorylation and the calcium response. To our knowledge, we are the first to report on the role of CCE in the mechanism of acute shear stress-induced NO response. In addition, our work highlights the importance of ATP autocrine signaling in shear stress-induced NO production.
Keywords: ATP; capacitative calcium entry; endothelial cells; nitric oxide; shear stress.
Conflict of interest statement
Conflicts of Interest: Dr. Andrews has nothing to disclose. Dr. Jaron reports grants from NIH, from NSF, during the conduct of the study. Dr. Buerk has nothing to disclose. Dr. Barbee reports grants from NIH, grants from NSF, during the conduct of the study.
Figures













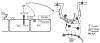
References
-
- Radomski MW, Palmer RM, Moncada S. Endogenous nitric oxide inhibits human platelet adhesion to vascular endothelium. Lancet. 1987;2:1057–1058. - PubMed
-
- Radomski MW, Vallance P, Whitley G, Foxwell N, Moncada S. Platelet adhesion to human vascular endothelium is modulated by constitutive and cytokine induced nitric oxide. Cardiovasc Res. 1993;27:1380–1382. - PubMed
-
- Tsao PS, Buitrago R, Chan JR, Cooke JP. Fluid flow inhibits endothelial adhesiveness. Nitric oxide and transcriptional regulation of VCAM-1. Circulation. 1996;94:1682–1689. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

