Cyclic avian mass mortality in the northeastern United States is associated with a novel orthomyxovirus
- PMID: 25392223
- PMCID: PMC4300652
- DOI: 10.1128/JVI.02019-14
Cyclic avian mass mortality in the northeastern United States is associated with a novel orthomyxovirus
Abstract
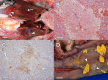
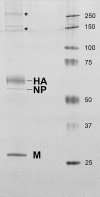
Since 1998, cyclic mortality events in common eiders (Somateria mollissima), numbering in the hundreds to thousands of dead birds, have been documented along the coast of Cape Cod, MA, USA. Although longitudinal disease investigations have uncovered potential contributing factors responsible for these outbreaks, detecting a primary etiological agent has proven enigmatic. Here, we identify a novel orthomyxovirus, tentatively named Wellfleet Bay virus (WFBV), as a potential causative agent of these outbreaks. Genomic analysis of WFBV revealed that it is most closely related to members of the Quaranjavirus genus within the family Orthomyxoviridae. Similar to other members of the genus, WFBV contains an alphabaculovirus gp64-like glycoprotein that was demonstrated to have fusion activity; this also tentatively suggests that ticks (and/or insects) may vector the virus in nature. However, in addition to the six RNA segments encoding the prototypical structural proteins identified in other quaranjaviruses, a previously unknown RNA segment (segment 7) encoding a novel protein designated VP7 was discovered in WFBV. Although WFBV shows low to moderate levels of sequence similarity to Quaranfil virus and Johnston Atoll virus, the original members of the Quaranjavirus genus, additional antigenic and genetic analyses demonstrated that it is closely related to the recently identified Cygnet River virus (CyRV) from South Australia, suggesting that WFBV and CyRV may be geographic variants of the same virus. Although the identification of WFBV in part may resolve the enigma of these mass mortality events, the details of the ecology and epidemiology of the virus remain to be determined.
Importance: The emergence or reemergence of viral pathogens resulting in large-scale outbreaks of disease in humans and/or animals is one of the most important challenges facing biomedicine. For example, understanding how orthomyxoviruses such as novel influenza A virus reassortants and/or mutants emerge to cause epidemic or pandemic disease is at the forefront of current global health concerns. Here, we describe the emergence of a novel orthomyxovirus, Wellfleet Bay virus (WFBV), which has been associated with cyclic large-scale bird die-offs in the northeastern United States. This initial characterization study provides a foundation for further research into the evolution, epidemiology, and ecology of newly emerging orthomyxoviruses, such as WFBV, and their potential impacts on animal and/or human health.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures




References
-
- Clark GM, O'Meara D, Van Weelden JW. 1958. An epizootic among eider ducks involving an acanthocephalid worm. J Wildlife Manage 22:204–205. doi:10.2307/3797332. - DOI
-
- Goudie RI, Robertson GJ, Reed A. 2000. Common eider (Somateria mollissima). InPoole AF, Gill FB (ed), The birds of North America, no. 177 The Academy of Natural Sciences, Washington, DC.
-
- Hario M, Lehtonen JT, Hollmén T. 1995. Role, if any, of the acanthocephalan worm Polymorphus minutus in common eider mortality. Suomen Riista 41:21–26.
-
- McCauley JW, Hongo S, Kaverin NV, Kochs G, Lamb RA, Matrosovich MN, Perez DR, Palese P, Presti RM, Rimstad E, Smith GJD. 2011. Family Orthomyxoviridae, p 749–761 InKing AMQ, Adams MJ, Carstens EB, Lefkowitz EJ (ed), Virus taxonomy: ninth report of the International Committee on Taxonomy of Viruses. Elsevier Academic Press, San Diego, CA.
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

