Use of diffusion magnetic resonance imaging to correlate the developmental changes in grape berry tissue structure with water diffusion patterns
- PMID: 25400688
- PMCID: PMC4232727
- DOI: 10.1186/1746-4811-10-35
Use of diffusion magnetic resonance imaging to correlate the developmental changes in grape berry tissue structure with water diffusion patterns
Erratum in
-
Erratum to: Use of diffusion magnetic resonance imaging to correlate the developmental changes in grape berry tissue structure with water diffusion patterns.Plant Methods. 2016 Jan 27;12:7. doi: 10.1186/s13007-016-0106-x. eCollection 2016. Plant Methods. 2016. PMID: 26823676 Free PMC article.
Abstract
Background: Over the course of grape berry development, the tissues of the berry undergo numerous morphological transformations in response to processes such as water and solute accumulation and cell division, growth and senescence. These transformations are expected to produce changes to the diffusion of water through these tissues detectable using diffusion magnetic resonance imaging (MRI). To assess this non-invasive technique diffusion was examined over the course of grape berry development, and in plant tissues with contrasting oil content.
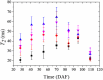
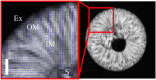
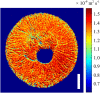
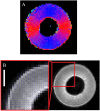
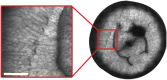
Results: In this study, the fruit of Vitis vinfera L. cv. Semillon at seven different stages of berry development, from four weeks post-anthesis to over-ripe, were imaged using diffusion tensor and transverse relaxation MRI acquisition protocols. Variations in diffusive motion between these stages of development were then linked to known events in the morphological development of the grape berry. Within the inner mesocarp of the berry, preferential directions of diffusion became increasingly apparent as immature berries increased in size and then declined as berries progressed through the ripening and senescence phases. Transverse relaxation images showed radial striation patterns throughout the sub-tissue, initiating at the septum and vascular systems located at the centre of the berry, and terminating at the boundary between the inner and outer mesocarp. This study confirms that these radial patterns are due to bands of cells of alternating width that extend across the inner mesocarp. Preferential directions of diffusion were also noted in young grape seed nucelli prior to their dehydration. These observations point towards a strong association between patterns of diffusion within grape berries and the underlying tissue structures across berry development. A diffusion tensor image of a post-harvest olive demonstrated that the technique is applicable to tissues with high oil content.
Conclusion: This study demonstrates that diffusion MRI is a powerful and information rich technique for probing the internal microstructure of plant tissues. It was shown that macroscopic diffusion anisotropy patterns correlate with the microstructure of the major pericarp tissues of cv. Semillon grape berries, and that changes in grape berry tissue structure during berry development can be observed.
Keywords: Development; Diffusion anisotropy; Diffusion tensor imaging; Grape berry; Nucellus; Nuclear magnetic resonance imaging; Olive; Seeds; Striation patterns; Vitis vinifera.
Figures











References
-
- Bernstein M, King K, Zhou X, Fong W. Handbook of MRI pulse sequences. Med Phys. 2005;32:1452. doi: 10.1118/1.1904597. - DOI
-
- Glidewell SM, Williamson B, Goodman BA, Chudek JA, Hunter G. An NMR microscopic study of grape (Vitis vinifera L.) Protoplasma. 1997;198:27–35. doi: 10.1007/BF01282128. - DOI
-
- Pope J, Jonas D, Walker R. Applications of NMR micro-imaging to the study of water, lipid, and carbohydrate distribution in grape berries. Protoplasma. 1993;173:177–186. doi: 10.1007/BF01379006. - DOI
-
- Van Der Weerd L, Claessens MMAE, Efdé C, Van As H. Nuclear magnetic resonance imaging of membrane permeability changes in plants during osmotic stress. Plant Cell Environ. 2002;25:1539–1549. doi: 10.1046/j.1365-3040.2002.00934.x. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources

