Optimization and critical evaluation of decellularization strategies to develop renal extracellular matrix scaffolds as biological templates for organ engineering and transplantation
- PMID: 25403742
- PMCID: PMC4276475
- DOI: 10.1111/ajt.12999
Optimization and critical evaluation of decellularization strategies to develop renal extracellular matrix scaffolds as biological templates for organ engineering and transplantation
Erratum in
-
Erratum.Am J Transplant. 2017 May;17(5):1429. doi: 10.1111/ajt.14250. Epub 2017 Mar 4. Am J Transplant. 2017. PMID: 28453228 No abstract available.
Abstract
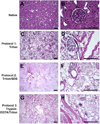
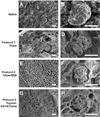
The ability to generate patient-specific cells through induced pluripotent stem cell (iPSC) technology has encouraged development of three-dimensional extracellular matrix (ECM) scaffolds as bioactive substrates for cell differentiation with the long-range goal of bioengineering organs for transplantation. Perfusion decellularization uses the vasculature to remove resident cells, leaving an intact ECM template wherein new cells grow; however, a rigorous evaluative framework assessing ECM structural and biochemical quality is lacking. To address this, we developed histologic scoring systems to quantify fundamental characteristics of decellularized rodent kidneys: ECM structure (tubules, vessels, glomeruli) and cell removal. We also assessed growth factor retention--indicating matrix biofunctionality. These scoring systems evaluated three strategies developed to decellularize kidneys (1% Triton X-100, 1% Triton X-100/0.1% sodium dodecyl sulfate (SDS) and 0.02% Trypsin-0.05% EGTA/1% Triton X-100). Triton and Triton/SDS preserved renal microarchitecture and retained matrix-bound basic fibroblast growth factor and vascular endothelial growth factor. Trypsin caused structural deterioration and growth factor loss. Triton/SDS-decellularized scaffolds maintained 3 h of leak-free blood flow in a rodent transplantation model and supported repopulation with human iPSC-derived endothelial cells and tubular epithelial cells ex vivo. Taken together, we identify an optimal Triton/SDS-based decellularization strategy that produces a biomatrix that may ultimately serve as a rodent model for kidney bioengineering.
Keywords: animal models: murine, bioengineering, kidney biology, stem cells; basic (laboratory) research/science; regenerative medicine; tissue/organ engineering.
© Copyright 2014 The American Society of Transplantation and the American Society of Transplant Surgeons.
Conflict of interest statement
The authors of this manuscript have no conflicts of interest to disclose as described by the American Journal of Transplantation.
Figures







References
-
- Gilbert TW, Sellaro TL, Badylak SF. Decellularization of tissues and organs. Biomaterials. 2006;27(19):3675–3683. - PubMed
-
- Rieder E, Kasimir MT, Silberhumer G, Seebacher G, Wolner E, Simon P, et al. Decellularization protocols of porcine heart valves differ importantly in efficiency of cell removal and susceptibility of the matrix to recellularization with human vascular cells. J Thorac Cardiovasc Surg. 2004;127(2):399–405. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

