Closure of the cytoplasmic gate formed by TM5 and TM11 during transport in the oxalate/formate exchanger from Oxalobacter formigenes
- PMID: 25409483
- PMCID: PMC4270380
- DOI: 10.1021/bi5012173
Closure of the cytoplasmic gate formed by TM5 and TM11 during transport in the oxalate/formate exchanger from Oxalobacter formigenes
Abstract
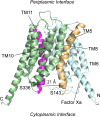
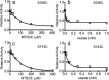
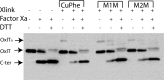
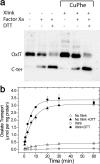
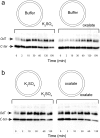
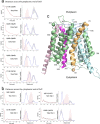
OxlT, the oxalate/formate exchanger of Oxalobacter formigenes, is a member of the major facilitator superfamily of transporters. In the present work, substrate (oxalate) was found to enhance the reactivity of the cysteine mutant S336C on the cytoplasmic end of helix 11 to methanethiosulfonate ethyl carboxylate. In addition, S336C is found to spontaneously cross-link to S143C in TM5 in either native or reconstituted membranes under conditions that support transport. Continuous wave EPR measurements are consistent with this result and indicate that positions 143 and 336 are in close proximity in the presence of substrate. These two residues are localized within helix interacting GxxxG-like motifs (G₁₄₀LASG₁₄₄ and S₃₃₆DIFG₃₄₀) at the cytoplasmic poles of TM5 and TM11. Pulse EPR measurements were used to determine distances and distance distributions across the cytoplasmic or periplasmic ends of OxlT and were compared with the predictions of an inside-open homology model. The data indicate that a significant population of transporter is in an outside-open configuration in the presence of substrate; however, each end of the transporter exhibits significant conformational heterogeneity, where both inside-open and outside-open configurations are present. These data indicate that TM5 and TM11, which form part of the transport pathway, transiently close during transport and that there is a conformational equilibrium between inside-open and outside-open states of OxlT in the presence of substrate.
Figures

Similar articles
-
The Structure and Function of OxlT, the Oxalate Transporter of Oxalobacter formigenes.J Membr Biol. 2015 Aug;248(4):641-50. doi: 10.1007/s00232-014-9728-y. Epub 2014 Sep 16. J Membr Biol. 2015. PMID: 25224873 Review.
-
Cysteine scanning mutagenesis of TM5 reveals conformational changes in OxlT, the oxalate transporter of Oxalobacter formigenes.Biochemistry. 2008 May 27;47(21):5709-17. doi: 10.1021/bi8001314. Epub 2008 May 2. Biochemistry. 2008. PMID: 18452311 Free PMC article.
-
Structure/function relationships in OxlT, the oxalate-formate transporter of oxalobacter formigenes. Assignment of transmembrane helix 11 to the translocation pathway.J Biol Chem. 2001 Mar 23;276(12):8753-60. doi: 10.1074/jbc.M008417200. Epub 2000 Dec 11. J Biol Chem. 2001. PMID: 11113128
-
Helix proximity in OxlT, the oalate:formate antiporter of oxalobacter formigenes. Cross-linking between TM2 and TM11.J Biol Chem. 2001 Sep 28;276(39):36681-6. doi: 10.1074/jbc.M106079200. Epub 2001 Jul 16. J Biol Chem. 2001. PMID: 11457863
-
Oxalobacter formigenes and its role in oxalate metabolism in the human gut.FEMS Microbiol Lett. 2004 Jan 15;230(1):1-7. doi: 10.1016/S0378-1097(03)00864-4. FEMS Microbiol Lett. 2004. PMID: 14734158 Review.
Cited by
-
Emulating proton-induced conformational changes in the vesicular monoamine transporter VMAT2 by mutagenesis.Proc Natl Acad Sci U S A. 2016 Nov 22;113(47):E7390-E7398. doi: 10.1073/pnas.1605162113. Epub 2016 Nov 7. Proc Natl Acad Sci U S A. 2016. PMID: 27821772 Free PMC article.
-
Crystal structure of bacterial haem importer complex in the inward-facing conformation.Nat Commun. 2016 Nov 10;7:13411. doi: 10.1038/ncomms13411. Nat Commun. 2016. PMID: 27830695 Free PMC article.
-
Atomic-level characterization of transport cycle thermodynamics in the glycerol-3-phosphate:phosphate antiporter.Nat Commun. 2015 Sep 29;6:8393. doi: 10.1038/ncomms9393. Nat Commun. 2015. PMID: 26417850 Free PMC article.
References
-
- Allison M. J.; Dawson K. A.; Mayberry W. R.; Foss J. G. (1985) Oxalobacter formigenes gen. nov., sp. nov.: oxalate-degrading anaerobes that inhabit the gastrointestinal tract. Arch. Microbiol. 141, 1–7. - PubMed
-
- Stewart C. S.; Duncan S. H.; Cave D. R. (2004) Oxalobacter formigenes and its role in oxalate metabolism in the human gut. FEMS Microbiol. Lett. 230, 1–7. - PubMed
-
- Maloney P. C. (1994) Bacterial transporters. Curr. Opin. Cell Biol. 6, 571–582. - PubMed
-
- Hirai T.; Heymann J. A.; Shi D.; Sarker R.; Maloney P. C.; Subramaniam S. (2002) Three-dimensional structure of a bacterial oxalate transporter. Nat. Struct. Biol. 9, 597–600. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

