Inositol-1,4,5-trisphosphate induced Ca2+ release and excitation-contraction coupling in atrial myocytes from normal and failing hearts
- PMID: 25416623
- PMCID: PMC4376424
- DOI: 10.1113/jphysiol.2014.283226
Inositol-1,4,5-trisphosphate induced Ca2+ release and excitation-contraction coupling in atrial myocytes from normal and failing hearts
Abstract
Key points: Impaired calcium (Ca(2+)) signalling is the main contributor to depressed ventricular contractile function and occurrence of arrhythmia in heart failure (HF). Here we report that in atrial cells of a rabbit HF model, Ca(2+) signalling is enhanced and we identified the underlying cellular mechanisms. Enhanced Ca(2+) transients (CaTs) are due to upregulation of inositol-1,4,5-trisphosphate receptor induced Ca(2+) release (IICR) and decreased mitochondrial Ca(2+) sequestration. Enhanced IICR, however, together with an increased activity of the sodium-calcium exchange mechanism, also facilitates spontaneous Ca(2+) release in form of arrhythmogenic Ca(2+) waves and spontaneous action potentials, thus enhancing the arrhythmogenic potential of atrial cells. Our data show that enhanced Ca(2+) signalling in HF provides atrial cells with a mechanism to improve ventricular filling and to maintain cardiac output, but also increases the susceptibility to develop atrial arrhythmias facilitated by spontaneous Ca(2+) release.
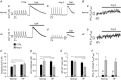
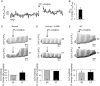
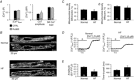
Abstract: We studied excitation-contraction coupling (ECC) and inositol-1,4,5-triphosphate (IP3)-dependent Ca(2+) release in normal and heart failure (HF) rabbit atrial cells. Left ventricular HF was induced by combined volume and pressure overload. In HF atrial myocytes diastolic [Ca(2+)]i was increased, action potential (AP)-induced Ca(2+) transients (CaTs) were larger in amplitude, primarily due to enhanced Ca(2+) release from central non-junctional sarcoplasmic reticulum (SR) and centripetal propagation of activation was accelerated, whereas HF ventricular CaTs were depressed. The larger CaTs were due to enhanced IP3 receptor-induced Ca(2+) release (IICR) and reduced mitochondrial Ca(2+) buffering, consistent with a reduced mitochondrial density and Ca(2+) uptake capacity in HF. Elementary IP3 receptor-mediated Ca(2+) release events (Ca(2+) puffs) were more frequent in HF atrial myoctes and were detected more often in central regions of the non-junctional SR compared to normal cells. HF cells had an overall higher frequency of spontaneous Ca(2+) waves and a larger fraction of waves (termed arrhythmogenic Ca(2+) waves) triggered APs and global CaTs. The higher propensity of arrhythmogenic Ca(2+) waves resulted from the combined action of enhanced IICR and increased activity of sarcolemmal Na(+)-Ca(2+) exchange depolarizing the cell membrane. In conclusion, the data support the hypothesis that in atrial myocytes from hearts with left ventricular failure, enhanced CaTs during ECC exert positive inotropic effects on atrial contractility which facilitates ventricular filling and contributes to maintaining cardiac output. However, HF atrial cells were also more susceptible to developing arrhythmogenic Ca(2+) waves which might form the substrate for atrial rhythm disorders frequently encountered in HF.
© 2014 The Authors. The Journal of Physiology © 2014 The Physiological Society.
Figures




Comment in
-
IP3 and Ca(2+) signals in the heart: boost them or bust them?J Physiol. 2015 Mar 15;593(6):1385-6. doi: 10.1113/jphysiol.2014.287987. J Physiol. 2015. PMID: 25772293 Free PMC article. No abstract available.
References
-
- Ai X, Curran JW, Shannon TR, Bers DM. Pogwizd SM. Ca2+/calmodulin-dependent protein kinase modulates cardiac ryanodine receptor phosphorylation and sarcoplasmic reticulum Ca2+ leak in heart failure. Circ Res. 2005;97:1314–1322. - PubMed
-
- Alpert JS, Petersen P. Godtfredsen J. Atrial fibrillation: natural history, complications, and management. Annu Rev Med. 1988;39:41–52. - PubMed
-
- Barth E, Stammler G, Speiser B. Schaper J. Ultrastructural quantitation of mitochondria and myofilaments in cardiac muscle from 10 different animal species including man. J Mol Cell Cardiol. 1992;24:669–681. - PubMed
-
- Bers DM, Pogwizd SM. Schlotthauer K. Upregulated Na/Ca exchange is involved in both contractile dysfunction and arrhythmogenesis in heart failure. Basic Res Cardiol. 2002;97(Suppl. 1):I36–42. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

