Global and phylogenetic distribution of quorum sensing signals, acyl homoserine lactones, in the family of Vibrionaceae
- PMID: 25419995
- PMCID: PMC4245543
- DOI: 10.3390/md12115527
Global and phylogenetic distribution of quorum sensing signals, acyl homoserine lactones, in the family of Vibrionaceae
Erratum in
-
Correction: Rasmussen, B.B., et al. Global and phylogenetic distribution of quorum sensing signals, acyl homoserine lactones, in the family of vibrionaceae. Mar. Drugs 2014, 12, 5527-5546.Mar Drugs. 2015 Mar 20;13(3):1548-51. doi: 10.3390/md13031548. Mar Drugs. 2015. PMID: 25803179 Free PMC article.
Abstract
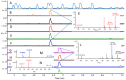
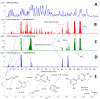
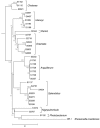
Bacterial quorum sensing (QS) and the corresponding signals, acyl homoserine lactones (AHLs), were first described for a luminescent Vibrio species. Since then, detailed knowledge has been gained on the functional level of QS; however, the abundance of AHLs in the family of Vibrionaceae in the environment has remained unclear. Three hundred and one Vibrionaceae strains were collected on a global research cruise and the prevalence and profile of AHL signals in this global collection were determined. AHLs were detected in 32 of the 301 strains using Agrobacterium tumefaciens and Chromobacterium violaceum reporter strains. Ethyl acetate extracts of the cultures were analysed by ultra-high performance liquid chromatography-high resolution mass spectrometry (MS) with automated tandem MS confirmation for AHLs. N-(3-hydroxy-hexanoyl) (OH-C6) and N-(3-hydroxy-decanoyl) (OH-C10) homoserine lactones were the most common AHLs found in 17 and 12 strains, respectively. Several strains produced a diversity of different AHLs, including N-heptanoyl (C7) HL. AHL-producing Vibrionaceae were found in polar, temperate and tropical waters. The AHL profiles correlated with strain phylogeny based on gene sequence homology, however not with geographical location. In conclusion, a wide range of AHL signals are produced by a number of clades in the Vibrionaceae family and these results will allow future investigations of inter- and intra-species interactions within this cosmopolitan family of marine bacteria.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

