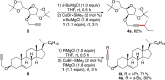
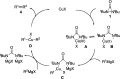
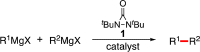Copper-catalyzed oxidative homo- and cross-coupling of Grignard reagents using diaziridinone
- PMID: 25420218
- PMCID: PMC4260645
- DOI: 10.1021/ol5030103
Copper-catalyzed oxidative homo- and cross-coupling of Grignard reagents using diaziridinone
Abstract
Transition-metal-catalyzed cross-coupling reactions are among the most powerful synthetic transformations. This paper describes an efficient copper-catalyzed homo- and cross-coupling of Grignard reagents with di-tert-butyldiaziridinone as oxidant under mild conditions, giving the coupling products in good to excellent yields. The reaction process has a broad substrate scope and is also effective for the C(sp)-C(sp(3)) coupling.
Figures
Similar articles
-
Highly efficient Cu(I)-catalyzed oxidation of alcohols to ketones and aldehydes with diaziridinone.Org Lett. 2013 Mar 1;15(5):992-5. doi: 10.1021/ol303431h. Epub 2013 Feb 15. Org Lett. 2013. PMID: 23413952 Free PMC article.
-
A facile copper(i)-catalyzed homo-coupling of indanone derivatives using diaziridinone under mild conditions.Org Biomol Chem. 2019 Jul 24;17(29):6998-7001. doi: 10.1039/c9ob00901a. Org Biomol Chem. 2019. PMID: 31304944
-
A facile copper(I)-catalyzed homocoupling of terminal alkynes to 1,3-diynes with diaziridinone under mild conditions.Org Biomol Chem. 2013 Nov 21;11(43):7451-4. doi: 10.1039/c3ob41621f. Org Biomol Chem. 2013. PMID: 24089251 Free PMC article.
-
Transition-metal-free oxidative coupling reactions for the formation of C-C and C-N bonds mediated by TEMPO and its derivatives.Chimia (Aarau). 2012;66(6):413-7. doi: 10.2533/chimia.2012.413. Chimia (Aarau). 2012. PMID: 22871285 Review.
-
Palladium-Catalyzed Organic Reactions Involving Hypervalent Iodine Reagents.Molecules. 2022 Jun 17;27(12):3900. doi: 10.3390/molecules27123900. Molecules. 2022. PMID: 35745020 Free PMC article. Review.
Cited by
-
Highly selective cross-coupling reactions of 1,1-dibromoethylenes with alkynylaluminums for the synthesis of aryl substituted conjugated enediynes and unsymmetrical 1,3-diynes.RSC Adv. 2022 May 3;12(21):13314-13318. doi: 10.1039/d2ra02127g. eCollection 2022 Apr 28. RSC Adv. 2022. PMID: 35520111 Free PMC article.
-
C(sp3)-C(sp3) bond formation via nickel-catalyzed deoxygenative homo-coupling of aldehydes/ketones mediated by hydrazine.Nat Commun. 2021 Jun 17;12(1):3729. doi: 10.1038/s41467-021-23971-7. Nat Commun. 2021. PMID: 34140496 Free PMC article.
-
Accessing Grignard Reluctant Aldehyde in 2-Oxoaldehyde by Organocuprates to Give [1,2] Addition and Oxidative Coupling Reactions.ACS Omega. 2022 Feb 1;7(6):5069-5078. doi: 10.1021/acsomega.1c06031. eCollection 2022 Feb 15. ACS Omega. 2022. PMID: 35187323 Free PMC article.
References
-
-
For leading reviews, see:
- Metal-Catalyzed Cross-Coupling Reactions; Diederich F., Stang P. J., Eds.; Wiley-VCH: Weinheim, 1998.
- Hassan J.; Sévignon M.; Gozzi C.; Schulz E.; Lemaire M. Chem. Rev. 2002, 102, 1359. - PubMed
- Littke A. F.; Fu G. C. Angew. Chem., Int. Ed. 2002, 41, 4176. - PubMed
- Metal-Catalyzed Cross-Coupling Reactions, 2nd Completely Revised and Enlarged ed.; de Meijere A., Diederich F., Eds.; Wiley-VCH: Weinheim, 2004.
- Nicolaou K. C.; Bulger P. G.; Sarlah D. Angew. Chem., Int. Ed. 2005, 44, 4442. - PubMed
- Corbet J.-P.; Mignani G. Chem. Rev. 2006, 106, 2651. - PubMed
- Kantchev E. A. B.; O’Brien C. J.; Organ M. G. Angew. Chem., Int. Ed. 2007, 46, 2768. - PubMed
- Jana R.; Pathak T. P.; Sigman M. S. Chem. Rev. 2011, 111, 1417. - PMC - PubMed
-
-
-
For leading reviews, see:
- Kauffmann T. Angew. Chem., Int. Ed. 1974, 13, 291.
- Iyoda M. Adv. Synth. Catal. 2009, 351, 984.
- Shao Z.; Peng F. Angew. Chem., Int. Ed. 2010, 49, 9566. - PubMed
- Liu C.; Jin L.; Lei A. Synlett 2010, 2527.
- Shi W.; Liu C.; Lei A. Chem. Soc. Rev. 2011, 40, 2761. - PubMed
- Liu C.; Zhang H.; Shi W.; Lei A. Chem. Rev. 2011, 111, 1780. - PubMed
-
-
- Bennett G. M.; Turner E. E. J. Chem. Soc. 1914, 1057.
- Gilman H.; Lichtenwalter M. J. Am. Chem. Soc. 1939, 61, 957.
- McKillop A.; Elsom L. F.; Taylor E. C. J. Am. Chem. Soc. 1968, 90, 2423.
- Lee J. I. J. Korean Chem. Soc. 2005, 49, 117.
-
-
For leading references on transition-metal-catalyzed homocoupling reactions of Grignard reagents with 1,2-dihaloethanes as oxidants, see:
- Nagano T.; Hayashi T. Org. Lett. 2005, 7, 491. - PubMed
- Cahiez G.; Chaboche C.; Mahuteau-Betzer F.; Ahr M. Org. Lett. 2005, 7, 1943. - PubMed
- Nagano T.; Hayashi T. Chem. Lett. 2005, 34, 1152.
- Zhou Z.; Xue W. J. Organomet. Chem. 2009, 694, 599.
- Kude K.; Hayase S.; Kawatsura M.; Itoh T. Heteroat. Chem. 2011, 22, 397.
-
-
-
For leading references on transition-metal-catalyzed homocoupling reactions of Grignard reagents with dioxygen as oxidant, see:
- Cahiez G.; Moyeux A.; Buendia J.; Duplais C. J. Am. Chem. Soc. 2007, 129, 13788. - PubMed
- Liu W.; Lei A. Tetrahedron Lett. 2008, 49, 610.
- Mayer M.; Czaplik W. M.; von Wangelin A. J. Synlett 2009, 2919.
- Aparna P. I.; Bhat B. R. J. Mol. Catal. A: Chem. 2012, 358, 73.
- Hua S.-K.; Hu Q.-P.; Ren J.; Zeng B.-B. Synthesis 2013, 45, 518.
- Bhat A. P. I.; Inam F.; Bhat B. R. Eur. J. Org. Chem. 2013, 7139.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources