Elevated hippocampal resting-state connectivity underlies deficient neurocognitive function in aging
- PMID: 25422457
- PMCID: PMC4267388
- DOI: 10.1073/pnas.1410233111
Elevated hippocampal resting-state connectivity underlies deficient neurocognitive function in aging
Abstract
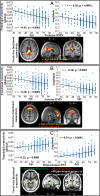
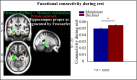
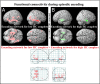
The brain is not idle during rest. Functional MRI (fMRI) studies have identified several resting-state networks, including the default mode network (DMN), which contains a set of cortical regions that interact with a hippocampus (HC) subsystem. Age-related alterations in the functional architecture of the DMN and HC may influence memory functions and possibly constitute a sensitive biomarker of forthcoming memory deficits. However, the exact form of DMN-HC alterations in aging and concomitant memory deficits is largely unknown. Here, using both task and resting data from 339 participants (25-80 y old), we have demonstrated age-related decrements in resting-state functional connectivity across most parts of the DMN, except for the HC network for which age-related elevation of connectivity between left and right HC was found along with attenuated HC-cortical connectivity. Elevated HC connectivity at rest, which was partly accounted for by age-related decline in white matter integrity of the fornix, was associated with lower cross-sectional episodic memory performance and declining longitudinal memory performance over 20 y. Additionally, elevated HC connectivity at rest was associated with reduced HC neural recruitment and HC-cortical connectivity during active memory encoding, which suggests that strong HC connectivity restricts the degree to which the HC interacts with other brain regions during active memory processing revealed by task fMRI. Collectively, our findings suggest a model in which age-related disruption in cortico-hippocampal functional connectivity leads to a more functionally isolated HC at rest, which translates into aberrant hippocampal decoupling and deficits during mnemonic processing.
Keywords: DMN; aging; episodic memory; hippocampus; resting state.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Modulation of effective connectivity in the default mode network at rest and during a memory task.Brain Connect. 2015 Feb;5(1):60-7. doi: 10.1089/brain.2014.0249. Epub 2014 Dec 29. Brain Connect. 2015. PMID: 25390185
-
The Longitudinal Trajectory of Default Mode Network Connectivity in Healthy Older Adults Varies As a Function of Age and Is Associated with Changes in Episodic Memory and Processing Speed.J Neurosci. 2018 Mar 14;38(11):2809-2817. doi: 10.1523/JNEUROSCI.3067-17.2018. Epub 2018 Feb 13. J Neurosci. 2018. PMID: 29440553 Free PMC article.
-
Default mode network connectivity indicates episodic memory capacity in mesial temporal lobe epilepsy.Epilepsia. 2013 May;54(5):809-18. doi: 10.1111/epi.12098. Epub 2013 Jan 29. Epilepsia. 2013. PMID: 23360362
-
Healthy aging by staying selectively connected: a mini-review.Gerontology. 2014;60(1):3-9. doi: 10.1159/000354376. Epub 2013 Sep 28. Gerontology. 2014. PMID: 24080587 Review.
-
A review of hippocampal activation in post-traumatic stress disorder.Psychophysiology. 2020 Jan;57(1):e13357. doi: 10.1111/psyp.13357. Epub 2019 Mar 4. Psychophysiology. 2020. PMID: 30829407 Review.
Cited by
-
Resting-state functional connectivity of the human hippocampus in periadolescent children: Associations with age and memory performance.Hum Brain Mapp. 2021 Aug 1;42(11):3620-3642. doi: 10.1002/hbm.25458. Epub 2021 May 12. Hum Brain Mapp. 2021. PMID: 33978276 Free PMC article.
-
Differential spatial working memory-related functional network reconfiguration in young and older adults.Netw Neurosci. 2024 Jul 1;8(2):395-417. doi: 10.1162/netn_a_00358. eCollection 2024. Netw Neurosci. 2024. PMID: 38952809 Free PMC article.
-
Tau deposition is associated with functional isolation of the hippocampus in aging.Nat Commun. 2019 Oct 25;10(1):4900. doi: 10.1038/s41467-019-12921-z. Nat Commun. 2019. PMID: 31653847 Free PMC article.
-
Altered hippocampal GABA and glutamate levels and uncoupling from functional connectivity in multiple sclerosis.Hippocampus. 2018 Nov;28(11):813-823. doi: 10.1002/hipo.23001. Hippocampus. 2018. PMID: 30069963 Free PMC article.
-
A Quantitative Data-Driven Analysis Framework for Resting-State Functional Magnetic Resonance Imaging: A Study of the Impact of Adult Age.Front Neurosci. 2021 Oct 20;15:768418. doi: 10.3389/fnins.2021.768418. eCollection 2021. Front Neurosci. 2021. PMID: 34744623 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

