Nitric oxide-mediated antioxidative mechanism in yeast through the activation of the transcription factor Mac1
- PMID: 25423296
- PMCID: PMC4244153
- DOI: 10.1371/journal.pone.0113788
Nitric oxide-mediated antioxidative mechanism in yeast through the activation of the transcription factor Mac1
Abstract
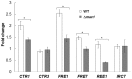
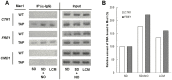
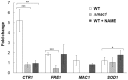
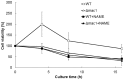
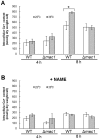
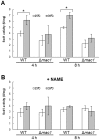
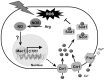
The budding yeast Saccharomyces cerevisiae possesses various defense mechanisms against environmental stresses that generate reactive oxygen species, leading to growth inhibition or cell death. Our recent study showed a novel antioxidative mechanism mediated by nitric oxide (NO) in yeast cells, but the mechanism underlying the oxidative stress tolerance remained unclear. We report here one of the downstream pathways of NO involved in stress-tolerance mechanism in yeast. Our microarray and real-time quantitative PCR analyses revealed that exogenous NO treatment induced the expression of genes responsible for copper metabolism under the control of the transcription factor Mac1, including the CTR1 gene encoding high-affinity copper transporter. Our ChIP analysis also demonstrated that exogenous NO enhances the binding of Mac1 to the promoter region of target genes. Interestingly, we found that NO produced under high-temperature stress conditions increased the transcription level of the CTR1 gene. Furthermore, NO produced during exposure to high temperature also increased intracellular copper content, the activity of Cu,Zn-superoxide dismutase Sod1, and cell viability after exposure to high-temperature in a manner dependent on Mac1. NO did not affect the expression of the MAC1 gene, indicating that NO activates Mac1 through its post-translational modification. Based on the results shown here, we propose a novel NO-mediated antioxidative mechanism that Mac1 activated by NO induces the CTR1 gene, leading to an increase in cellular copper level, and then Cu(I) activates Sod1. This is the first report to unveil the mechanism of NO-dependent antioxidative system in yeast.
Conflict of interest statement
Figures
Similar articles
-
Transcriptional activation in yeast in response to copper deficiency involves copper-zinc superoxide dismutase.J Biol Chem. 2009 Jan 2;284(1):404-413. doi: 10.1074/jbc.M807027200. Epub 2008 Oct 31. J Biol Chem. 2009. PMID: 18977757 Free PMC article.
-
The essential liaison of two copper proteins: the Cu-sensing transcription factor Mac1 and the Cu/Zn superoxide dismutase Sod1 in Saccharomyces cerevisiae.Curr Genet. 2023 Feb;69(1):41-53. doi: 10.1007/s00294-022-01258-8. Epub 2022 Dec 1. Curr Genet. 2023. PMID: 36456733
-
Synergy of Hir1, Ssn6, and Snf2 global regulators is the functional determinant of a Mac1 transcriptional switch in S. cerevisiae copper homeostasis.Curr Genet. 2019 Jun;65(3):799-816. doi: 10.1007/s00294-019-00935-5. Epub 2019 Jan 28. Curr Genet. 2019. PMID: 30689022
-
Copper-regulatory domain involved in gene expression.Prog Nucleic Acid Res Mol Biol. 1998;58:165-95. doi: 10.1016/s0079-6603(08)60036-7. Prog Nucleic Acid Res Mol Biol. 1998. PMID: 9308366 Review.
-
Copper metabolism in Saccharomyces cerevisiae: an update.Biometals. 2021 Feb;34(1):3-14. doi: 10.1007/s10534-020-00264-y. Epub 2020 Oct 30. Biometals. 2021. PMID: 33128172 Review.
Cited by
-
Metallothionein Cup1 attenuates nitrosative stress in the yeast Saccharomyces cerevisiae.Microb Cell. 2023 Jul 10;10(8):170-177. doi: 10.15698/mic2023.08.802. eCollection 2023 Aug 7. Microb Cell. 2023. PMID: 37545644 Free PMC article.
-
Identification and Functional Analysis of GTP Cyclohydrolase II in Candida glabrata in Response to Nitrosative Stress.Front Microbiol. 2022 Mar 2;13:825121. doi: 10.3389/fmicb.2022.825121. eCollection 2022. Front Microbiol. 2022. PMID: 35308400 Free PMC article.
-
Alternative oxidase gene induced by nitric oxide is involved in the regulation of ROS and enhances the resistance of Pleurotus ostreatus to heat stress.Microb Cell Fact. 2021 Jul 19;20(1):137. doi: 10.1186/s12934-021-01626-y. Microb Cell Fact. 2021. PMID: 34281563 Free PMC article.
-
Methylarginine efflux in nutrient-deprived yeast mitigates disruption of nitric oxide synthesis.Amino Acids. 2023 Feb;55(2):215-233. doi: 10.1007/s00726-022-03220-x. Epub 2022 Dec 1. Amino Acids. 2023. PMID: 36454288
-
MAC Family Transcription Factors Enhance the Tolerance of Mycelia to Heat Stress and Promote the Primordial Formation Rate of Pleurotus ostreatus.J Fungi (Basel). 2023 Dec 27;10(1):13. doi: 10.3390/jof10010013. J Fungi (Basel). 2023. PMID: 38248923 Free PMC article.
References
-
- Woo J, Yang K, Kim S, Blank L, Park J (2014) High temperature stimulates acetic acid accumulation and enhances the growth inhibition and ethanol production by Saccharomyces cerevisiae under fermenting. Appl Microbiol Biotechnol: DOI 10.1007/s00253–014–5691–x. doi:10.1007/s00253-014-5691-x. - PubMed
-
- Pérez-Gallardo RV, Briones LS, Díaz-Pérez AL, Gutiérrez S, Rodríguez-Zavala JS, et al. (2013) Reactive oxygen species production induced by ethanol in Saccharomyces cerevisiae increases because of a dysfunctional mitochondrial iron-sulfur cluster assembly system. FEMS Yeast Res 13: 804–819 10.1111/1567-1364.12090 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

