The role of galectin-3 and galectin-3-binding protein in venous thrombosis
- PMID: 25428218
- PMCID: PMC4357586
- DOI: 10.1182/blood-2014-04-569939
The role of galectin-3 and galectin-3-binding protein in venous thrombosis
Abstract
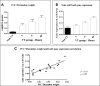
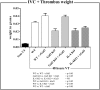
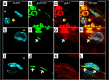
Galectin-3-binding protein (gal3bp) and its receptor/ligand, galectin-3 (gal3), are secreted proteins that initiate signaling cascades in several diseases, and recent human proteomic data suggest they may play a role in venous thrombosis (VT). We hypothesized that gal3bp and gal3 may promote VT. Using a mouse stasis model of VT, we found that gal3bp and gal3 were localized on vein wall, red blood cells, platelets, and microparticles, whereas leukocytes expressed gal3 only. Gal3 was dramatically increased during early VT and gal3bp:gal3 colocalized in the leukocyte/endothelial cell interface, where leukocytes were partially attached to the vein wall. Thrombus size correlated with elevated gal3 and interleukin-6 (IL-6) vein wall levels. Recombinant gal3 promoted VT and increased vein wall IL-6 mRNA. Although recombinant gal3 restored the VT size in gal3(-/-) mice, it had no effect on IL6(-/-) mice, suggesting that gal3:gal3bp promotes VT through IL-6. Moreover, significantly fewer activated neutrophils were present in the gal3(-/-) vein walls. In a group of human patients, elevated circulating gal3bp correlated with acute VT. In conclusion, gal3bp:gal3 play a critical role in VT, likely via IL-6 and PMN-mediated thrombotic mechanisms, and may be a potential biomarker in human VT.
© 2015 by The American Society of Hematology.
Figures





References
-
- Wakefield TW, McLafferty RB, Lohr JM, Caprini JA, Gillespie DL, Passman MA Executive Committee of the American Venous Forum. Call to action to prevent venous thromboembolism. J Vasc Surg. 2009;49(6):1620–1623. - PubMed
-
- Linkins LA, Choi PT, Douketis JD. Clinical impact of bleeding in patients taking oral anticoagulant therapy for venous thromboembolism: a meta-analysis. Ann Intern Med. 2003;139(11):893–900. - PubMed
-
- Woo HJ, Lotz MM, Jung JU, Mercurio AM. Carbohydrate-binding protein 35 (Mac-2), a laminin-binding lectin, forms functional dimers using cysteine 186. J Biol Chem. 1991;266(28):18419–18422. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

