Highly pathogenic avian influenza virus infection in chickens but not ducks is associated with elevated host immune and pro-inflammatory responses
- PMID: 25431115
- PMCID: PMC4246556
- DOI: 10.1186/s13567-014-0118-3
Highly pathogenic avian influenza virus infection in chickens but not ducks is associated with elevated host immune and pro-inflammatory responses
Abstract
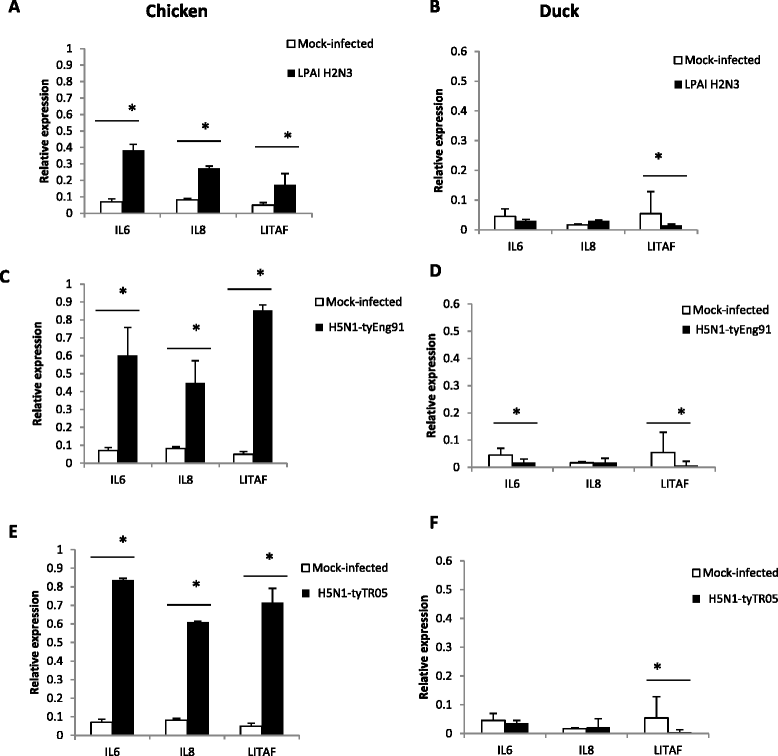
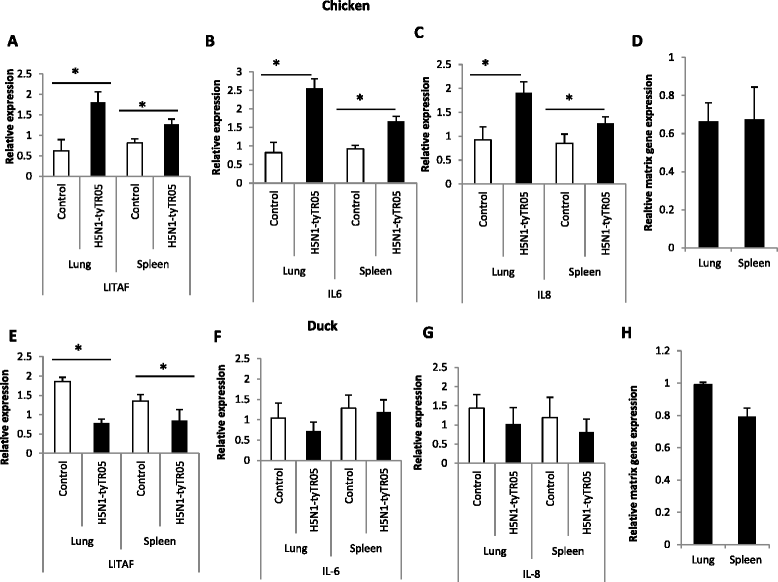
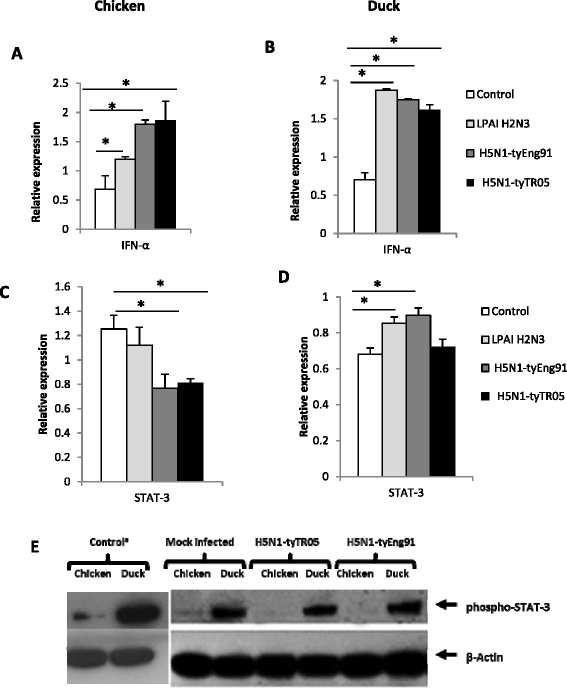
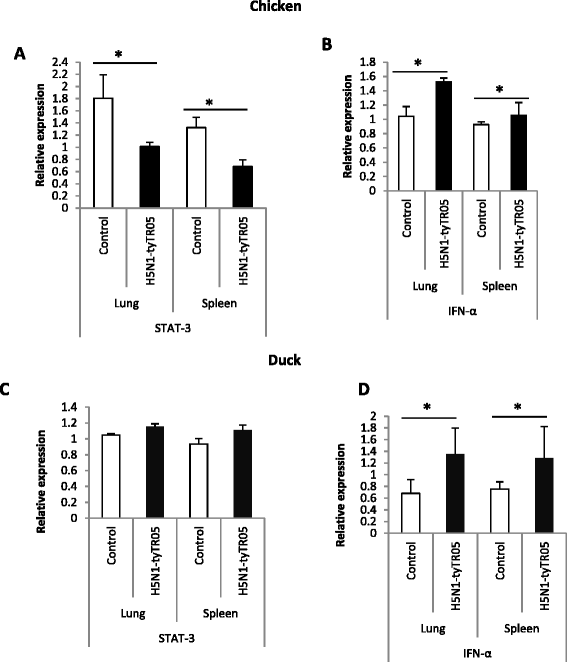
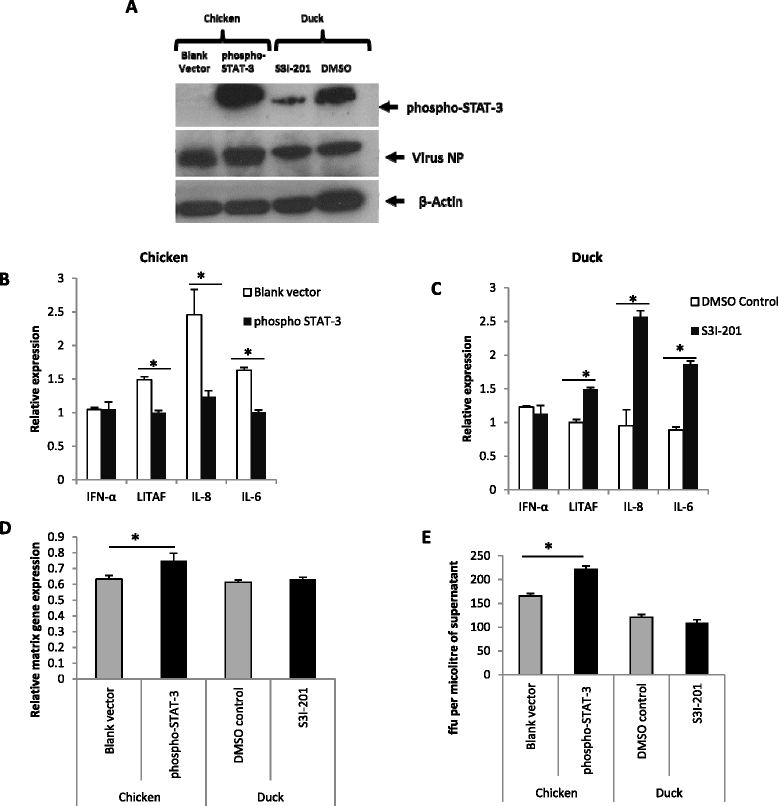
Highly pathogenic avian influenza (HPAI) H5N1 viruses cause severe infection in chickens at near complete mortality, but corresponding infection in ducks is typically mild or asymptomatic. To understand the underlying molecular differences in host response, primary chicken and duck lung cells, infected with two HPAI H5N1 viruses and a low pathogenicity avian influenza (LPAI) H2N3 virus, were subjected to RNA expression profiling. Chicken cells but not duck cells showed highly elevated immune and pro-inflammatory responses following HPAI virus infection. HPAI H5N1 virus challenge studies in chickens and ducks corroborated the in vitro findings. To try to determine the underlying mechanisms, we investigated the role of signal transducer and activator of transcription-3 (STAT-3) in mediating pro-inflammatory response to HPAIV infection in chicken and duck cells. We found that STAT-3 expression was down-regulated in chickens but was up-regulated or unaffected in ducks in vitro and in vivo following H5N1 virus infection. Low basal STAT-3 expression in chicken cells was completely inhibited by H5N1 virus infection. By contrast, constitutively active STAT-3 detected in duck cells was unaffected by H5N1 virus infection. Transient constitutively-active STAT-3 transfection in chicken cells significantly reduced pro-inflammatory response to H5N1 virus infection; on the other hand, chemical inhibition of STAT-3 activation in duck cells increased pro-inflammatory gene expression following H5N1 virus infection. Collectively, we propose that elevated pro-inflammatory response in chickens is a major pathogenicity factor of HPAI H5N1 virus infection, mediated in part by the inhibition of STAT-3.
Figures
Similar articles
-
Efficacy of a Recombinant Turkey Herpesvirus H5 Vaccine Against Challenge With H5N1 Clades 1.1.2 and 2.3.2.1 Highly Pathogenic Avian Influenza Viruses in Domestic Ducks (Anas platyrhynchos domesticus).Avian Dis. 2016 Mar;60(1):22-32. doi: 10.1637/11282-091615-Reg.1. Avian Dis. 2016. PMID: 26953940
-
Duck MDA5 functions in innate immunity against H5N1 highly pathogenic avian influenza virus infections.Vet Res. 2014 Jun 18;45(1):66. doi: 10.1186/1297-9716-45-66. Vet Res. 2014. PMID: 24939427 Free PMC article.
-
Genetic characterization and pathogenicity assessment of highly pathogenic H5N1 avian influenza viruses isolated from migratory wild birds in 2011, South Korea.Virus Res. 2011 Sep;160(1-2):305-15. doi: 10.1016/j.virusres.2011.07.003. Epub 2011 Jul 12. Virus Res. 2011. PMID: 21782862
-
Vaccination of domestic ducks against H5N1 HPAI: a review.Virus Res. 2013 Dec 5;178(1):21-34. doi: 10.1016/j.virusres.2013.07.012. Epub 2013 Aug 3. Virus Res. 2013. PMID: 23916865 Review.
-
Innate Immune Responses to Avian Influenza Viruses in Ducks and Chickens.Vet Sci. 2019 Jan 10;6(1):5. doi: 10.3390/vetsci6010005. Vet Sci. 2019. PMID: 30634569 Free PMC article. Review.
Cited by
-
Comparative Investigation of Coincident Single Nucleotide Polymorphisms Underlying Avian Influenza Viruses in Chickens and Ducks.Biology (Basel). 2023 Jul 7;12(7):969. doi: 10.3390/biology12070969. Biology (Basel). 2023. PMID: 37508399 Free PMC article.
-
A review of H5Nx avian influenza viruses.Ther Adv Vaccines Immunother. 2019 Feb 22;7:2515135518821625. doi: 10.1177/2515135518821625. eCollection 2019. Ther Adv Vaccines Immunother. 2019. PMID: 30834359 Free PMC article. Review.
-
Resistant and susceptible chicken lines show distinctive responses to Newcastle disease virus infection in the lung transcriptome.BMC Genomics. 2017 Dec 28;18(1):989. doi: 10.1186/s12864-017-4380-4. BMC Genomics. 2017. PMID: 29281979 Free PMC article.
-
Differential gene expression reveals host factors for viral shedding variation in mallards (Anas platyrhynchos) infected with low-pathogenic avian influenza virus.J Gen Virol. 2022 Mar;103(3):10.1099/jgv.0.001724. doi: 10.1099/jgv.0.001724. J Gen Virol. 2022. PMID: 35353676 Free PMC article.
-
Comparative Investigation of Gene Regulatory Processes Underlying Avian Influenza Viruses in Chicken and Duck.Biology (Basel). 2022 Jan 29;11(2):219. doi: 10.3390/biology11020219. Biology (Basel). 2022. PMID: 35205087 Free PMC article.
References
-
- Thitithanyanont A, Engering A, Uiprasertkul M, Ekchariyawat P, Wiboon-Ut S, Kraivong R, Limsalakpetch A, Kum-Arb U, Yongvanitchit K, Sa-Ard-Iam N, Rukyen P, Mahanonda R, Kawkitinarong K, Auewarakul P, Utaisincharoen P, Sirisinha S, Mason CJ, Fukuda MM, Pichyangkul S. Antiviral immune responses in H5N1-infected human lung tissue and possible mechanisms underlying the hyperproduction of interferon-inducible protein IP-10. Biochem Biophys Res Commun. 2010;398:752–758. doi: 10.1016/j.bbrc.2010.07.017. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

