Telodendrimer nanocarrier for co-delivery of paclitaxel and cisplatin: A synergistic combination nanotherapy for ovarian cancer treatment
- PMID: 25453973
- PMCID: PMC4312198
- DOI: 10.1016/j.biomaterials.2014.10.044
Telodendrimer nanocarrier for co-delivery of paclitaxel and cisplatin: A synergistic combination nanotherapy for ovarian cancer treatment
Abstract
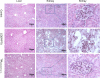
Cisplatin (CDDP) and paclitaxel (PTX) are two established chemotherapeutic drugs used in combination for the treatment of many cancers, including ovarian cancer. We have recently developed a three-layered linear-dendritic telodendrimer micelles (TM) by introducing carboxylic acid groups in the adjacent layer via "thio-ene" click chemistry for CDDP complexation and conjugating cholic acids via peptide chemistry in the interior layer of telodendrimer for PTX encapsulation. We hypothesize that the co-delivery of low dosage PTX with CDDP could act synergistically to increase the treatment efficacy and reduce their toxic side effects. This design allowed us to co-deliver PTX and CDDP at various drug ratios to ovarian cancer cells. The in vitro cellular assays revealed strongest synergism in anti-tumor effects when delivered at a 1:2 PTX/CDDP loading ratio. Using the SKOV-3 ovarian cancer xenograft mouse model, we demonstrate that our co-encapsulation approach resulted in an efficient tumor-targeted drug delivery, decreased cytotoxic effects and stronger anti-tumor effect, when compared with free drug combination or the single loading TM formulations.
Keywords: Cisplatin; Combination chemotherapy; Drug delivery; Ovarian cancer; Paclitaxel; Synergism.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures







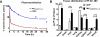



References
-
- Rowinsky EK, Gilbert MR, McGuire WP, Noe DA, Grochow LB, Forastiere AA, et al. Sequences of taxol and cisplatin: a phase I and pharmacologic study. J Clin Oncol. 1991;9:1692–703. - PubMed
-
- Milross CG, Peters LJ, Hunter NR, Mason KA, Milas L. Sequence-dependent antitumor activity of paclitaxel (taxol) and cisplatin in vivo. Int J Cancer. 1995;62:599–604. - PubMed
-
- Sacchi A, Gasparri A, Gallo-Stampino C, Toma S, Curnis F, Corti A. Synergistic antitumor activity of cisplatin, paclitaxel, and gemcitabine with tumor vasculature-targeted tumor necrosis factor-alpha. Clin Cancer Res. 2006;12:175–82. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

