Design of the randomized, controlled sequential staged treatment of emphysema with upper lobe predominance (STEP-UP) study
- PMID: 25467378
- PMCID: PMC4265492
- DOI: 10.1186/1471-2466-14-190
Design of the randomized, controlled sequential staged treatment of emphysema with upper lobe predominance (STEP-UP) study
Abstract
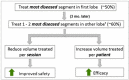
Background: An innovative approach to lung volume reduction (LVR) for emphysema is introduced in the design of the Sequential Segmental Treatment of Emphysema with Upper Lobe Predominance (STEP-UP) trial where vapour ablation is administered bilaterally over the course of two sessions and is used to target only the most diseased upper lobe segments. By dividing the procedure into two sessions, there is potential to increase the total volume treated per patient but reduce volume treated and energy delivered per session. This is expected to correlate with improvements in vapour ablation's safety and efficacy profiles.
Methods: The STEP-UP trial is a randomized, controlled, open-label, 12 month study of patients with upper lobe predominant emphysema (ULPE). The trial compares patients receiving standard medical management alone against patients receiving bilateral vapour ablation in addition to standard medical management. An intended sixty nine subjects will be randomized at a 2:1 (treatment arm:control arm) ratio. Inclusion criteria include a forced expiratory volume in 1 second (FEV1) between 20% and 45% predicted, total lung capacity > 100% predicted, residual volume > 150% predicted, marked dyspnea scoring ≥ 2 on the modified Medical Research Council (mMRC) scale, and PaCO2 ≤ 50 mm Hg. The primary endpoints are the change in FEV1 %predicted and St. George Respiratory Questionnaire (SGRQ) score between the treatment and control arm at 12 months. Adverse events will be monitored as secondary endpoints along with other efficacy outcomes at 6 and 12 months.
Discussion: Vapour ablation can reduce lung volume in the presence of collateral ventilation (CV). Due to this ability, it can be used to target specifically the more diseased segments of each upper lobe. Safety and efficacy outcomes are expected to improve by considering which segments to treat along with the volume treated per session and per patient.
Trial registration: ClinicalTrials.gov NCT01719263.
Figures



References
-
- Mathers C, Doris Ma F, Boerma JT. The Global Burden of Disease: 2004 Update. Geneva, Switzerland: World Health Organization; 2008.
-
- Fishman A, Martinez F, Naunheim K, Piantadosi S, Wise R, Ries A, Weinmann G, Wood DE, National Emphysema Treatment Trial Research Group A randomized trial comparing lung-volume-reduction surgery with medical therapy for severe emphysema. N Engl J Med. 2003;348(21):2059–2073. doi: 10.1056/NEJMoa030287. - DOI - PubMed
-
- DeCamp MM, Blackstone EH, Naunheim KS, Krasna MJ, Wood DE, Meli YM, McKenna RJ, Jr, NETT Research Group Patient and surgical factors influencing air leak after lung volume reduction surgery: lessons learned from the national emphysema treatment trial. Ann Thorac Surg. 2006;82(1):197–207. doi: 10.1016/j.athoracsur.2006.02.050. - DOI - PubMed
-
- Naunheim KS, Wood DE, Mohsenifar Z, Sternberg AL, Criner GJ, DeCamp MM, Deschamps CC, Martinez FJ, Sciurba FC, Tonascia J, Fishman AP. Long-term follow-up of patients receiving lung-volume-reduction surgery versus medical therapy for severe emphysema by the national emphysema treatment trial research group. Ann Thorac Surg. 2006;82(2):431–443. doi: 10.1016/j.athoracsur.2006.05.069. - DOI - PubMed
Pre-publication history
-
- The pre-publication history for this paper can be accessed here:http://www.biomedcentral.com/1471-2466/14/190/prepub
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

