Semaglutide, a once-weekly human GLP-1 analog, does not reduce the bioavailability of the combined oral contraceptive, ethinylestradiol/levonorgestrel
- PMID: 25475122
- PMCID: PMC4418331
- DOI: 10.1002/jcph.443
Semaglutide, a once-weekly human GLP-1 analog, does not reduce the bioavailability of the combined oral contraceptive, ethinylestradiol/levonorgestrel
Abstract
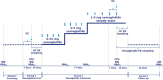
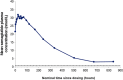
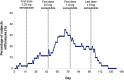
The effect of semaglutide, a once-weekly human glucagon-like peptide-1 (GLP-1) analog in development for type 2 diabetes (T2D), on the bioavailability of a combined oral contraceptive was investigated. Postmenopausal women with T2D (n = 43) on diet/exercise ± metformin received ethinylestradiol (0.03 mg)/levonorgestrel (0.15 mg) once daily for 8 days before (semaglutide-free) and during (steady-state 1.0 mg) semaglutide treatment (subcutaneous once weekly; dose escalation: 0.25 mg 4 weeks; 0.5 mg 4 weeks; 1.0 mg 5 weeks). Bioequivalence of oral contraceptives was established if 90%CI for the ratio of pharmacokinetic parameters during semaglutide steady-state and semaglutide-free periods was within prespecified limits (0.80-1.25). The bioequivalence criterion was met for ethinylestradiol area under the curve (AUC0-24 h ) for semaglutide steady-state/semaglutide-free; 1.11 (1.06-1.15). AUC0-24 h was 20% higher for levonorgestrel at semaglutide steady-state vs. semaglutide-free (1.20 [1.15-1.26]). Cmax was within bioequivalence criterion for both contraceptives. Reductions (mean ± SD) in HbA1c (-1.1 ± 0.6%) and weight (-4.3 ± 3.1 kg) were observed. Semaglutide pharmacokinetics were compatible with once-weekly dosing; the semaglutide dose and dose-escalation regimen were well tolerated. Adverse events, mainly gastrointestinal, were mild to moderate in severity. Asymptomatic increases in mean amylase and lipase were observed. Three subjects had elevated alanine aminotransferase levels ≥3x the upper limit of normal during semaglutide/oral contraceptive coadministration, which were reported as adverse events, but resolved during follow-up. Semaglutide did not reduce the bioavailability of ethinylestradiol and levonorgestrel.
Keywords: GLP-1; ethinylestradiol; levonorgestrel; once weekly; semaglutide; type 2 diabetes.
© 2015 The Authors. The Journal of Clinical Pharmacology Published by Wiley Periodicals, Inc. on behalf of American College of Clinical Pharmacology.
Figures


References
-
- Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006;368(9548):1696–1705. - PubMed
-
- Naslund E, Barkeling B, King N, et al. Energy intake and appetite are suppressed by glucagon-like peptide-1 (GLP-1) in obese men. Int J Obes Relat Metab Disord. 1999;23(3):304–311. - PubMed
-
- Willms B, Werner J, Holst JJ, Orskov C, Creutzfeldt W, Nauck MA. Gastric emptying, glucose responses, and insulin secretion after a liquid test meal: effects of exogenous glucagon-like peptide-1 (GLP-1)-(7–36) amide in type 2 (noninsulin-dependent) diabetic patients. J Clin Endocrinol Metab. 1996;81(1):327–332. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

